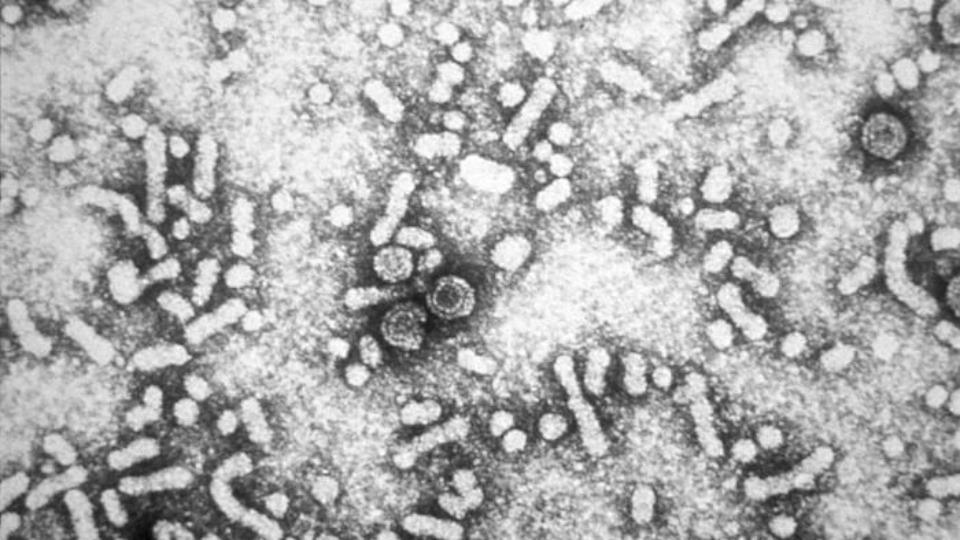
GSK eyes October FDA verdict on chronic hep B drug
GSK's antisense-based drug for chronic hepatitis B (CHB), bepirovirsen, has started a speedy FDA review that should deliver a verdict by 26th October.
Newsletters and Deep Dive digital magazine

GSK's antisense-based drug for chronic hepatitis B (CHB), bepirovirsen, has started a speedy FDA review that should deliver a verdict by 26th October.

Which companies are working with the government to implement the US-UK pharmaceutical trade agreement?

The FDA dropped a surprise Friday announcement that three more National Priority Vouchers would be given to a trio of psychedelics companies.

AbbVie's bid to bring a rapid and shorter-acting botulinum toxin product to the US market has been knocked back by the FDA.

Regeneron's Otarmeni is the first gene therapy for congenital hearing loss to be cleared by the FDA, and will be made available for free in the US.
Editor's Picks
Newsletters and Deep Dive
digital magazine