
Axsome breaks new ground in Alzheimer's with Auvelity OK
Axsome's Auvelity has become the first non-antipsychotic therapy for agitation in patients with Alzheimer's disease to be approved by the FDA.
Newsletters and Deep Dive digital magazine

Axsome's Auvelity has become the first non-antipsychotic therapy for agitation in patients with Alzheimer's disease to be approved by the FDA.

UCB's Rystiggo can be used by the NHS as a backup treatment for people with the rare autoimmune and neuromuscular disease gMG, says NICE.

Patients in the EU with the ultra-rare disease WHIM syndrome now have their first approved treatment, Norgine's Xolremdi.

The Association of the British Pharmaceutical Industry (ABPI)'s flagship annual conference took place on Thursday, 23rd April.
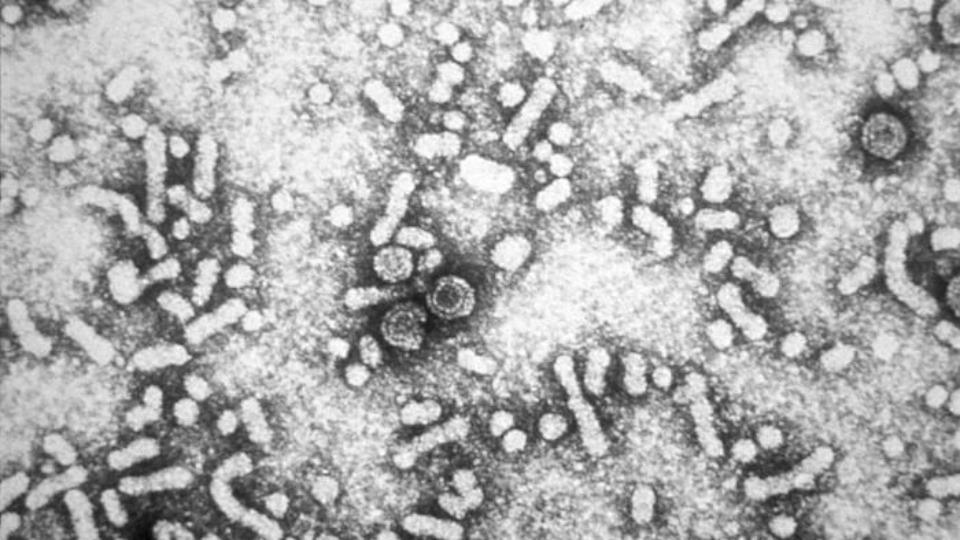
GSK's antisense-based drug for chronic hepatitis B (CHB), bepirovirsen, has started a speedy FDA review that should deliver a verdict by 26th October.
Editor's Picks
Newsletters and Deep Dive
digital magazine