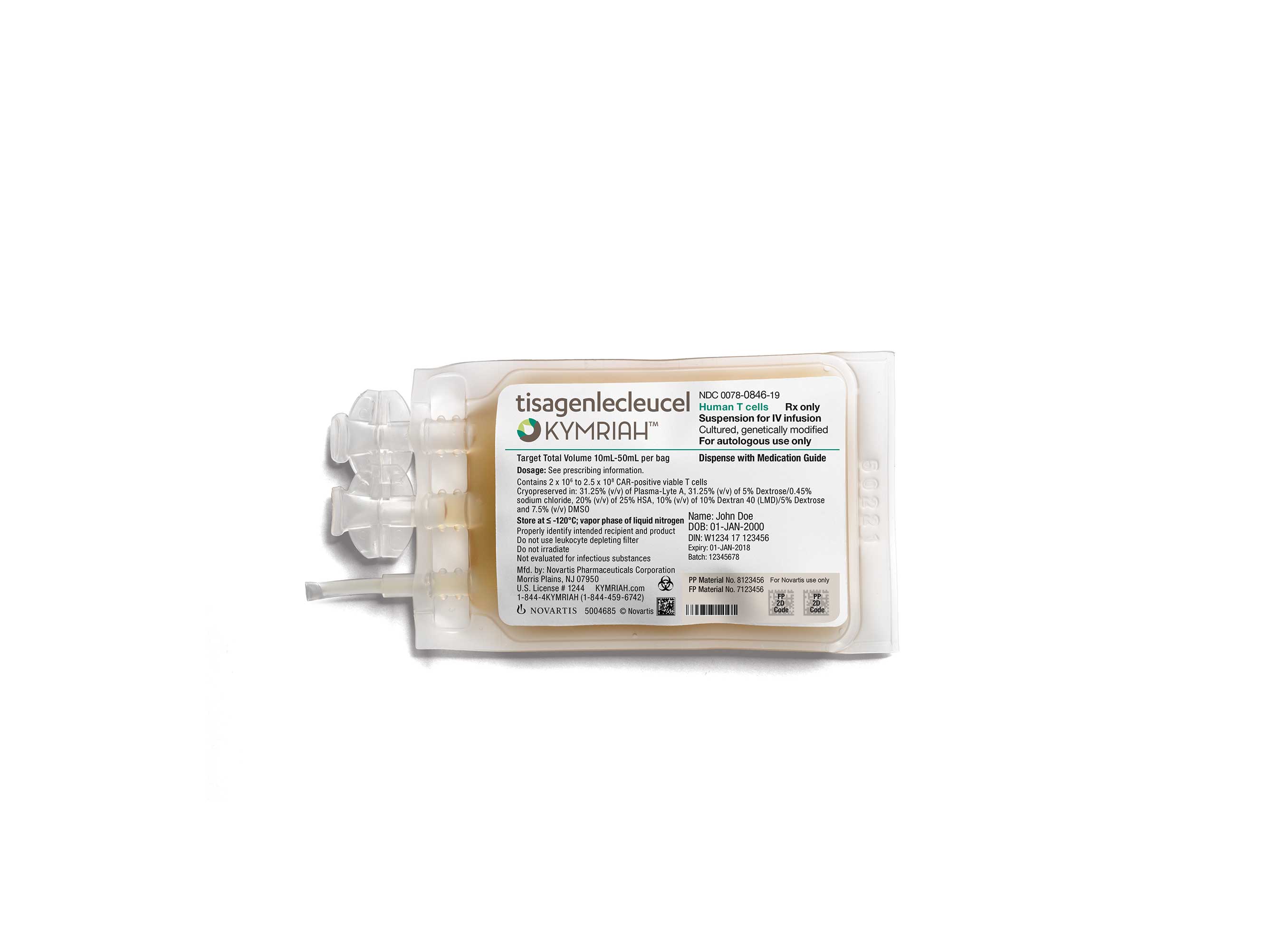
Gilead Sciences' push into oncology is paying off, as Veklur...
Gilead Sciences' investment in oncology has started to provide a solid return, with sales of cancer therapies breaching the $500 million threshold for the first time in the second quarter.<