Pharma’s search loophole is a preview of what comes next
A new research letter on Ozempic’s pay-per-click (PPC) strategy highlights a widening regulatory gap in pharmaceutical advertising. For marketers, the findings offer a rare view into how major drug companies use search to reach consumers in ways that regulators have not fully addressed. The US Food and Drug Administration (FDA) requires drug advertising to be “truthful, balanced, and accurate” while disclosing key information. But PPC search ads challenge the limits of that framework.
The study analysed two years of search ads linked to Ozempic.com. An estimated $7.5m was spent on 15,000+ keywords, generating 2.4m website visits. Many of these keywords were not related to diabetes, Ozempic’s approved use. Instead, a large share targeted searches about weight loss and competitors.
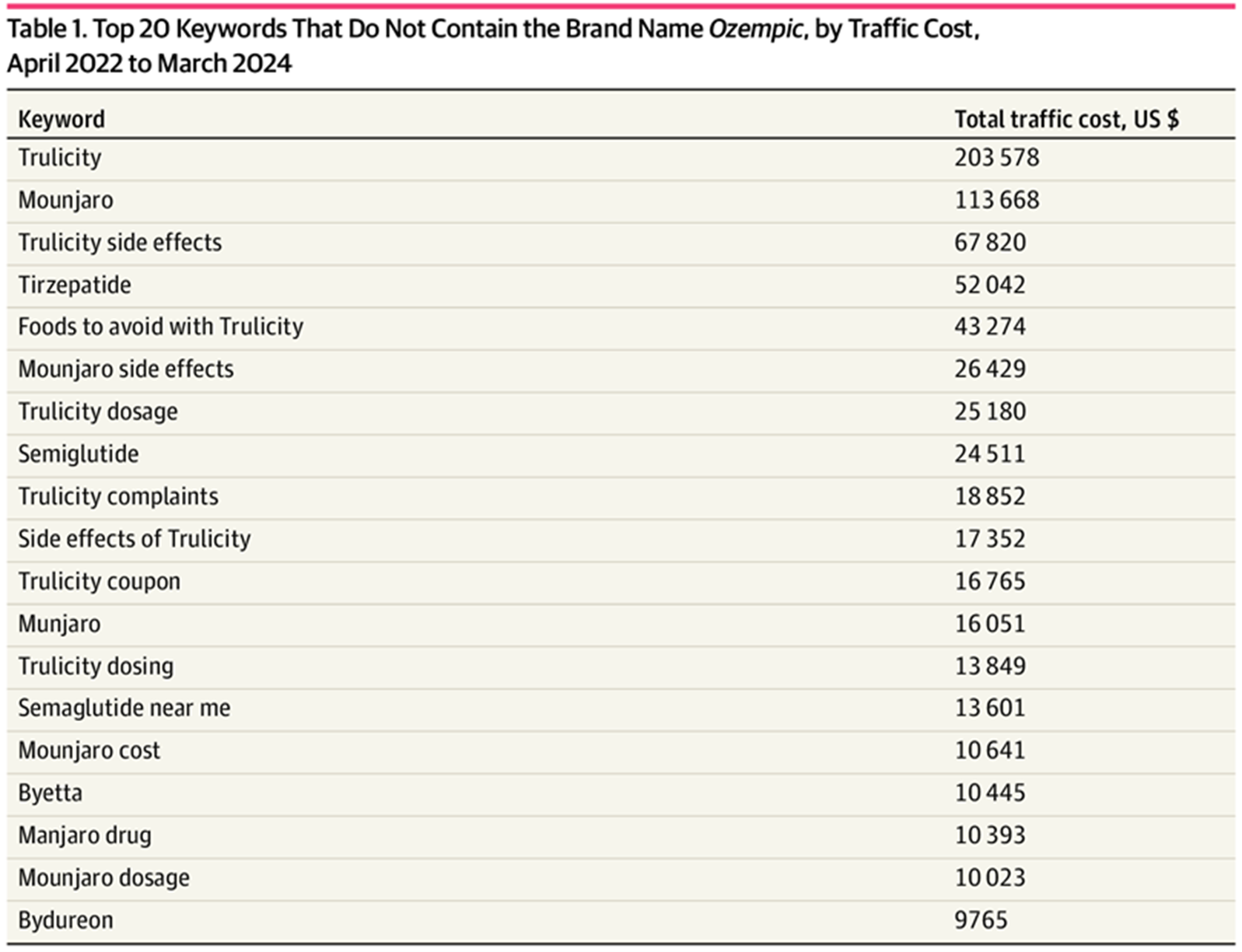
The table below shows that more than 23% of keywords did not mention Ozempic at all, including misspellings.
Top 20 Keywords That Do Not Contain the Brand Name Ozempic, by Traffic Cost, April 2022 to March 2024
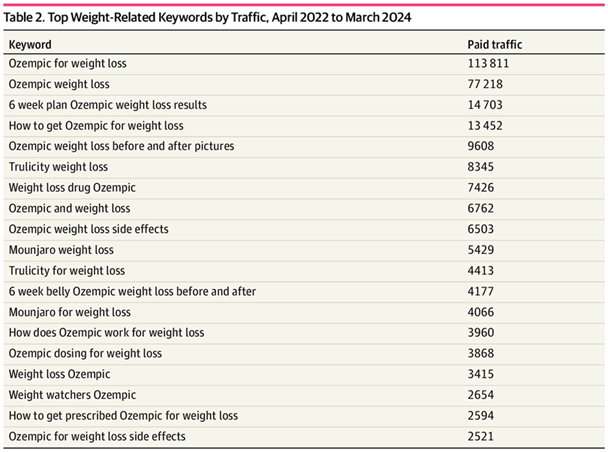
Additionally, 1729 keywords (11%) contained weight. Keywords related to weight generated over 358,000 website visits, accounting for 14% of all paid keyword searches.
Top Weight-Related Keywords by Traffic, April 2022 to March 2024
The regulatory tension lies in how these ads function. Drug makers are prohibited from promoting medications for unapproved uses. Yet, PPC ads often contain so little text that they avoid direct claims. Many consist only of a brand name, a link, and limited copy. This allows companies to promote their drugs for non-FDA-approved uses by appearing alongside searches related to off-label use.
If an ad does not make a direct claim, regulators have little basis to treat it as off-label promotion. But the ad still reaches consumers searching off-label topics. People searching weight-related terms were served ads for Ozempic even though the drug “is not FDA approved for weight loss”. The targeting is driven by keyword bidding, not explicit messaging. As a result, PPC acts as an indirect channel that can shape consumer interest without violating the law.
Brand control and search platforms
For marketers, this is a significant loophole. It shows how keyword targeting can reach high-intent audiences in regulated categories, and brands can enter a conversation they cannot officially join. It also shows how search platforms give brands control over visibility in moments when consumers are seeking information, not advertising.
FDA regulations governing drug promotion were developed around traditional media formats – print ads, television spots, and full web pages – where companies can provide the required risk and safety disclosures alongside any claims. Although the agency has since issued guidance for manufacturers’ use of social-media platforms, that guidance does not address SEM. As a result, PPC search ads sit in a regulatory gray zone. Because these ads contain so little text, they can avoid making any explicit therapeutic claim; and without a claim, the FDA’s fair-balance and disclosure requirements are not triggered. This allows pharmaceutical brands to bid on and appear alongside searches related to off-label topics as long as the ad directs users to a landing page that includes full legal disclosures. The result is a loophole in which SEM becomes a powerful, lightly regulated channel that operates ahead of existing FDA rules.
This creates interesting tension at a time when pharma advertising is undergoing a hard reset. It shows how digital ad tech can create opportunities for marketers long before official rules catch up, and why search intent data is becoming central to marketing strategies.
If search is the present example of how intent-based advertising can outpace regulation, then, what’s next?
That question became much less theoretical a few weeks ago when OpenAI said it would introduce ads into ChatGPT; a move many had assumed was coming, but had not been formally announced. The cultural and business implications were clear when Anthropic’s Claude used its Super Bowl spots to mock OpenAI’s ChatGPT, positioning an ad-free experience as a competitive advantage. The tagline: “Ads are coming to AI. But not to Claude.” Daily active users of Claude rose 11% after the game, site visits jumped 6.5%, and it surpassed competitors Meta, Google Gemini, and OpenAI in the top 10 free apps on the App Store.
The response marks a shift in the perception of AI from a conversational tool to an advertising channel.
Search monetises intent through keywords, and AI surfaces intent through conversation. A person might ask, “My doctor says I have Type 2 diabetes, and I want to understand medication options, what are people taking?” In an ad-supported environment, that conversation becomes a contextual signal that could trigger sponsored ad placement alongside the chat.
A regulatory grey area
FDA oversight is built around claims for pharmaceuticals: what is said about a drug, how risks are disclosed, and whether the messaging is fair and balanced. Paid search disrupted that model by separating targeting from content. Brands can bid on weight loss-related or competitor queries without making off-label statements in the ad copy. The ad appears in proximity to the inquiry while required disclosures sit on the landing page, so the copy is compliant and the targeting does the work.
This regulatory grey area could potentially be amplified with AI-driven search. Instead of bidding on keywords, advertisers may eventually bid on conversational context. Instead of appearing next to a query, a brand could appear alongside a chat about symptoms or treatments. In this case, same as search, the promotional copy remains compliant, but the targeting would again sit outside the traditional claim-based framework that governs pharma advertising.
Ozempic’s search strategy demonstrates how brands can influence through targeting just as much as promotional language. As AI companies begin testing ads, the same dynamic that reshaped search may extend into highly personalised conversational environments. The future of pharma marketing may depend even less on a brand’s direct claims and more on the intent signals they are allowed to monetise.
About the author

Eleanor Carey is a media strategist with AMS, offering expertise in comprehensive media planning, advanced audience targeting, and modern measurement methodologies that integrate analytics, testing, and real-time optimisation to drive stronger results across digital and traditional channels.