Return on Engagement (ROE): A new framework to better capture patient engagement value

In the life sciences industry, we measure outcomes as value: revenue, pipeline velocity, time to market, shareholder return. Often, we ask for short-term Return on Investment (ROI).
But here’s the uncomfortable question: when it comes to patient engagement, are we looking at it all in the wrong way? Engagement does not behave like sales. Patient engagement is not a transactional function, yet, we keep applying short-term ROI logic to long-term relational work. In the context of patient engagement, short-term ROI doesn’t make much sense.
This is not just a question of metrics, but of priorities. If the effectiveness of patient engagement is measured only by immediate financial return, we undermine the long-term prospects of creating products that meet the needs of patients.
Return on Engagement (ROE) is a call to action to better capture the multifaceted value of patient engagement.
ROE: A new framework for thinking about patient engagement
ROE in patient engagement is a concept that emerged during a keynote panel discussion at the recent World Patient Centricity Congress in London. While discussing ROI related to patient engagement, patient advocate Danielle Drachmann pointed out that the life sciences industry has been missing the point. How we have thought about patient engagement has been all wrong – we need to change our perspective from short-term financial gains to long-term value.
While ROE has been applied in other business contexts, such as marketing, Drachmann called for patient engagement to be considered in terms of ROE – the long-term value generated when patient engagement positively influences decision-making, improves understanding, accelerates innovation, reduces risk, and builds long-term trust in the patient community. In this model, patient engagement is not project-based, but rather an institutionalised, integrated, and central aspect of the life sciences industry.

ROI asks a fair question: “Did this make us money?” But that’s not the only question that matters. ROE looks at a bigger picture: “Did patient engagement help align our long-term strategy with the needs of the patient community?”
While ROI asks “What did patient engagement deliver this quarter?”, ROE asks “Did we build community trust that makes future innovation possible?”
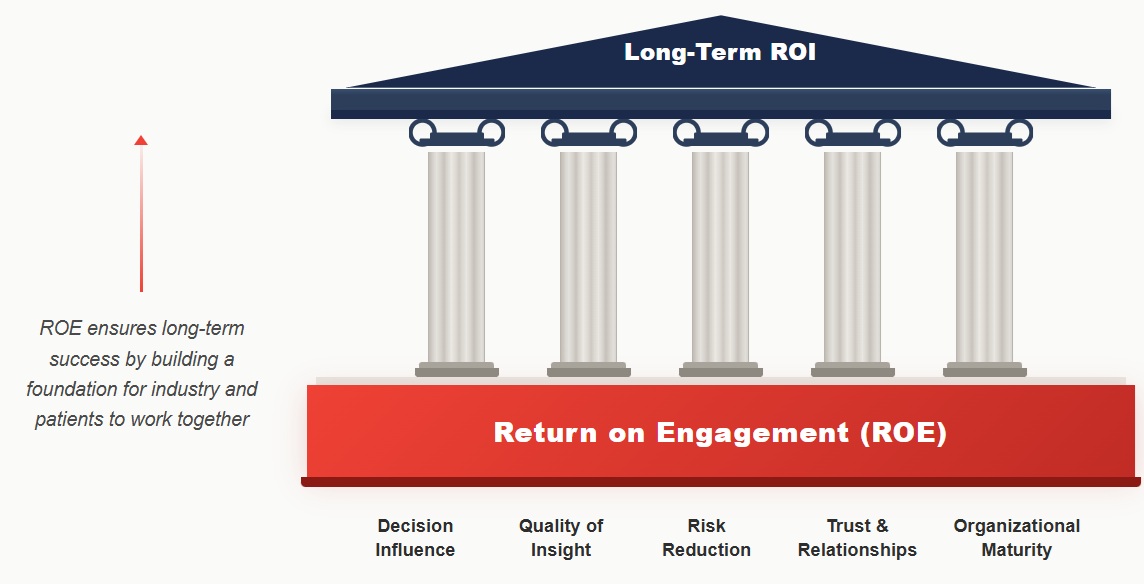
While ROI tracks transactions, ROE builds a foundation for industry and patients to work together to meet the needs of the patient community, ensuring long-term success.
How ROE works – and how it can be tracked
ROE measures the influence of patient engagement, rather than its effect on short-term profitability. ROE can be tracked by documenting how patient input influences decisions throughout the product lifecycle. This can be done using a structured patient engagement impact log that tracks where and how patient perspectives sparked new ideas, led to new collaborations, changed research questions and/or protocol design, shaped endpoint selection, informed portfolio priorities, or strengthened access and communication strategies. Some main dimensions of ROE and examples of possible ROE metrics are shown in the figure below.

ROE and ROI are complementary concepts
Patient engagement should be viewed as a long-term investment. When embedded properly and early, patient engagement delivers sustainable, long-term ROI by:
- Reducing development costs and shortening timelines
- Strengthening regulatory credibility and relationships
- Increasing the likelihood of reimbursement success
- Accelerating product launches and improving launch performance
If patient engagement is being done right – meaning, it is embedded as an integrated part of the business – it will be difficult to calculate its ROI using traditional methods. Nonetheless, when patient engagement is approached strategically, rather than transactionally, long-term ROI follows.
ROE considers whether:
- Endpoints reflect lived reality
- Patients feel safe enough to speak up and challenge assumptions
- Patient communities are willing to participate – and to remain engaged
- Teams can shift from informing patients to partnering with them
- Disseminations about the research are clear and accessible
- Trust is built between industry and the patient community
- Innovation is created to reflect patients’ real needs
ROE is smart business
Engaging with patients should never just be considered a “feel-good moment”, where we listen to patients’ stories and then get back to business as usual. We should consider patients and caregivers experts with valuable information critical to shaping strategy.
Patient engagement, when institutionalised, reduces blind spots in high-stakes, often billion-dollar decisions. Framed this way, ROE is an infrastructural approach to strengthen decision quality and ensure long-term value.
When industry and impacted patient communities are true partners, the result is stronger science, reduced risk, and more credible innovation. This is not just good practice – it’s smart business.
About the authors

Danielle Drachmann, MSc, is an award-winning rare disease advocate and global patient engagement leader. She is a senior patient partnering manager at the PPD clinical research business of Thermo Fisher Scientific, where she supports all of the company’s patient partnering activities. She is also the founder and CEO of Ketotic Hypoglycemia International, focused on helping families impacted by idiopathic ketotic hypoglycaemia, driving research with families from the patient community. Recipient of the 2022 Young Patient Advocate Award at the EURORDIS Black Pearl Awards, she also serves on the Advisory Board of the International Society for Patient Engagement Professionals (ISPEP), supporting the global advancement and professionalisation of patient engagement.

Phil Leventhal, PhD, is a senior manager at the PPD clinical research business of Thermo Fisher Scientific, where he leads the Publications and Scientific Communications team. With more than 20 years of experience in medical writing and scientific communications, he combines deep practical expertise with strategic planning to deliver effective communication solutions. Leventhal is a strong advocate for writing for the reader and for using storytelling in scientific communication to make complex science clear, relevant, and meaningful. He is passionate about patient authorship and engagement and, since 2024, has collaborated with Danielle Drachman on multiple initiatives to advance patient involvement in research and publications.

Stephanie Terrey, MSc, is a senior director of patient insights research at the PPD clinical research business of Thermo Fisher Scientific, where she leads teams designing and executing qualitative and quantitative research to inform patient-centred clinical development strategies. With more than 15 years of experience in healthcare research, she combines deep methodological expertise with strategic leadership to deliver insights that improve study design, recruitment, and patient engagement. Terrey is a strong advocate for elevating the patient voice and translating real-world experiences into actionable solutions that enhance trial success and inclusivity.












