Corcept gets FDA OK for first-in-class ovarian cancer drug
Almost four months ahead of schedule, Corcept Therapeutics' selective glucocorticoid receptor antagonist (SGRA) relacorilant has been cleared for sale in the US as a treatment for ovarian, fallopian tube, or primary peritoneal cancer.
The early FDA approval – under the Lifyorli brand name – covers use of the first-in-class, oral therapy, in combination with nab-paclitaxel, for adults with platinum-resistant forms of these cancers who have previously been treated with one to three prior systemic regimens.
It's also a major boost for Corcept after relacorilant was turned down by the US regulator for patients with hypertension secondary to Cushing syndrome (hypercortisolism) at the end of 2025.
The FDA's decision – well ahead of the action date of 11th July – comes on the back of the ROSELLA study, which showed a significant improvement in both progression-free survival (PFS) and overall survival (OS) compared to nab-paclitaxel alone. The data showed a 35% reduction in the risk of death, with patients taking Lifyorli living a median of 16 months versus around 12 months with nab-paclitaxel monotherapy.
"Lifyorli is positioned to become a new standard-of-care treatment," said Texas Oncology's Rob Coleman, who is special advisor to the president of the GOG Foundation, which represents patients with gynaecologic malignancies.
"Having a new treatment for this advanced, recurrent disease will provide clinicians with a compelling option to help patients with this extremely difficult-to-treat cancer."
Along with its ovarian cancer programme, Corcept is already running trials of relacorilant in endometrial, cervical, pancreatic, and prostate cancers, and chief executive Joseph Belanoff said the approval is a "first step" for the product with "much more to explore with this new mode of treatment."
Lifyorli featured in Clarivate's Drugs to Watch in 2026 listing as a biomarker-agnostic alternative for ovarian cancer for patients who aren't eligible for targeted therapies – even if the report's estimate of $150 million in G7 country sales by 2031 is fairly modest.
Other analysts are more optimistic, with UBS analyst Ashwani Verma estimating peak sales of $550 million, held back a little by a contraindication for patients on steroid medications, according to a Reuters report. That would be a major addition to Corcept's revenues, which were just over $760 million last year.
News of the approval prompted a 65% spike in Corcept's share price, which was hit hard by the FDA rejection in Cushing syndrome. While not returning to its 52-week high of more than $117, it is now running above $40, hiking the company's valuation to around $4.3 billion.
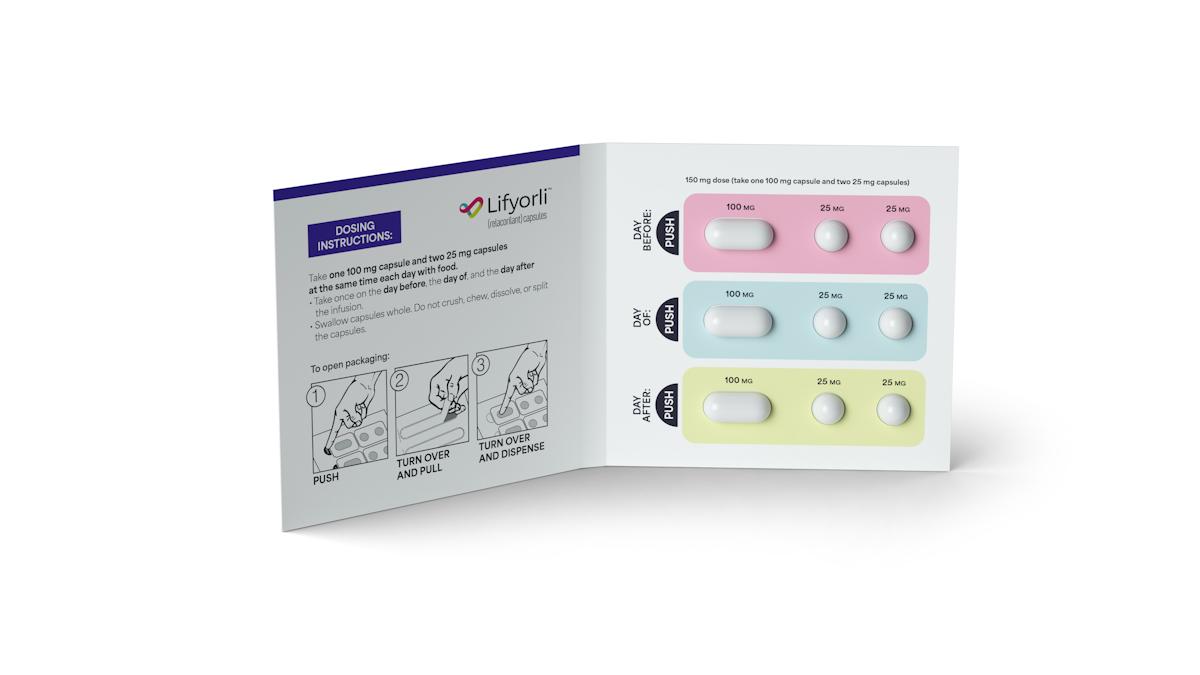
Corcept has set a wholesale price of $37,900 for a four-week cycle of Lifyorli and plans to launch it within the next few days. The treatment is administered as a 150 mg oral dose a day before, on the day, and a day after each infusion with nab-paclitaxel, which is given once a week.












