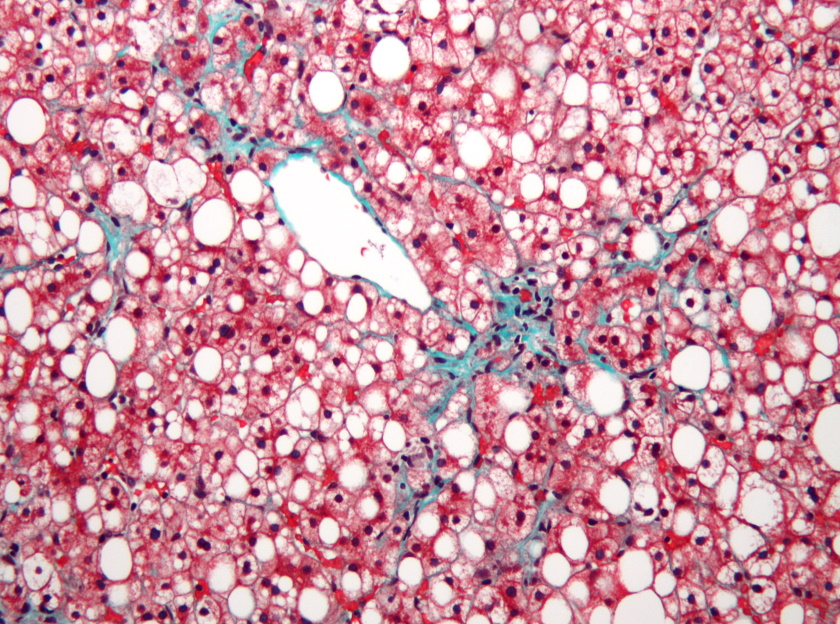
News

Madrigal Pharma is first over the MASH finish line
Madrigal Pharma has become the first drugmaker to claim FDA approval for a drug to treat metabolic dysfunction-associated steatohepatitis (MASH), an elusive target that ha