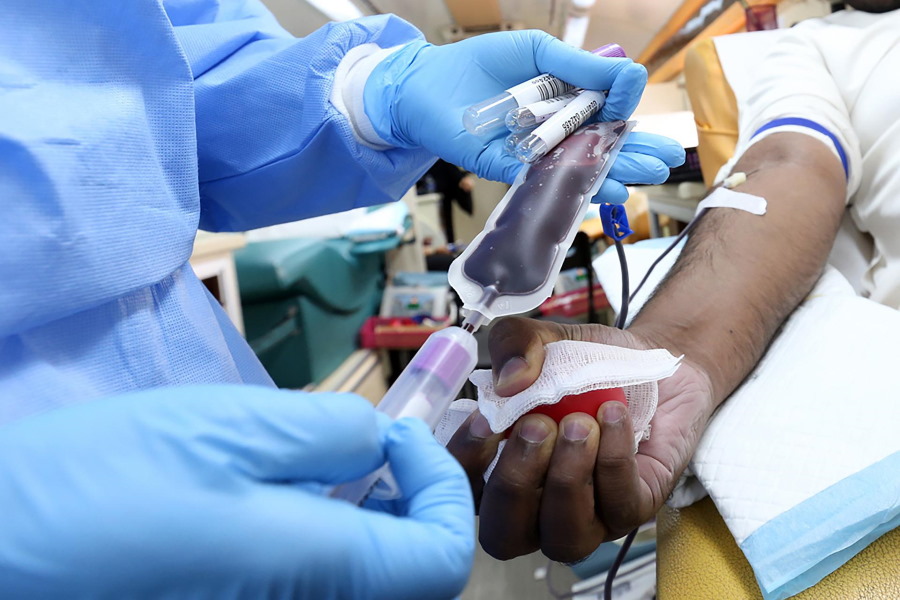
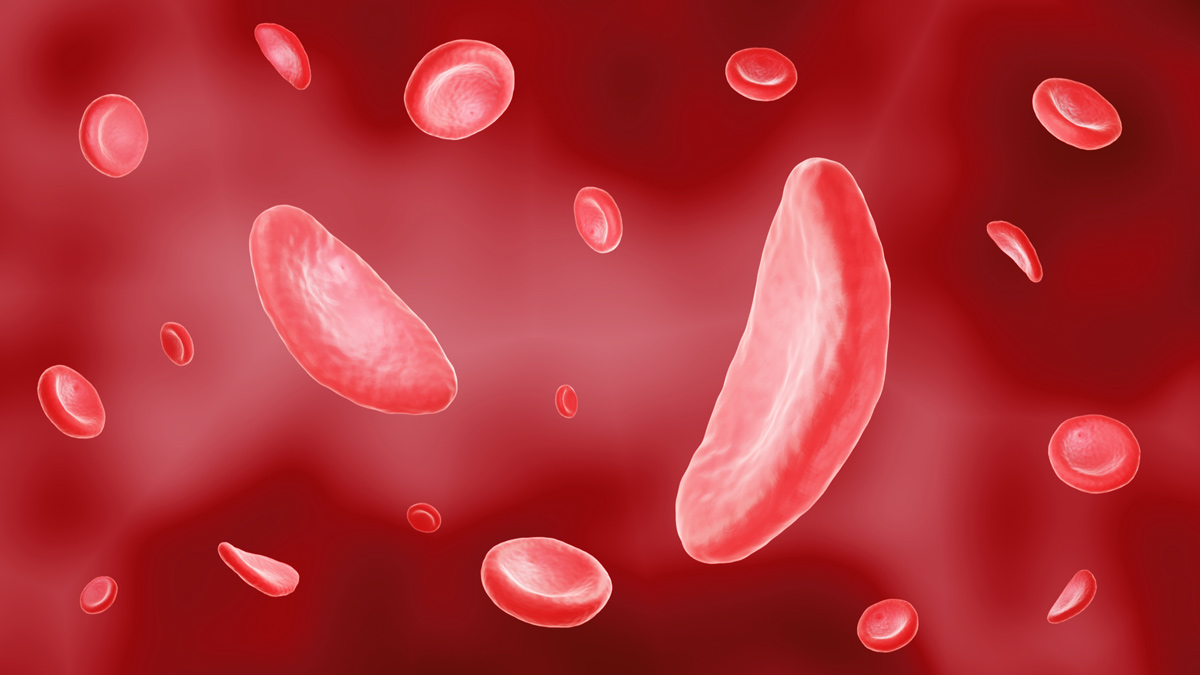
Vertex, CRISPR prep filing for gene-editing blood disorder t...
Vertex Pharma and partner CRISPR Therapeutics will start a rolling marketing application in the US for their gene-editing drug for sickle cell disease (SCD) and beta thalassaemia later this