Vaxxas announces CEPI partnership to advance needle-free mRNA vaccine patches
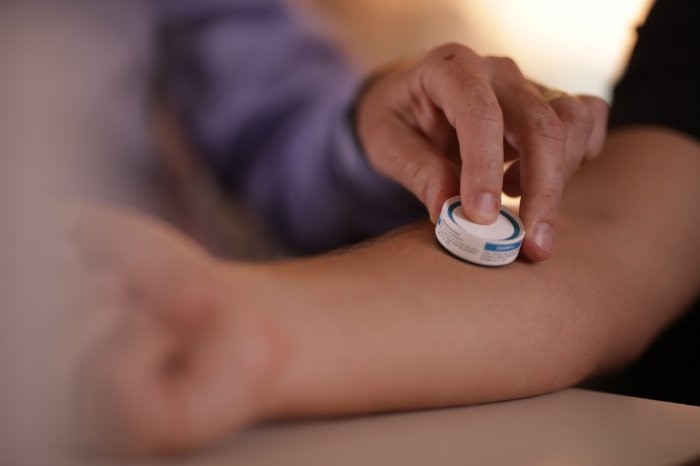
Biotech Vaxxas has announced a partnership agreement with the 2017-established Coalition for Epidemic Preparedness Innovations (CEPI), in order to advance the development of Vaxxas’ needle-free vaccine-patch delivery technology with preclinical testing of an mRNA vaccine patch.
CEPI is a partnership between public, private, philanthropic, and civil organisations to develop vaccines against future epidemics and pandemics.
The agreement, worth $4.3 million, is the first to be announced as part of CEPI’s call in January 2022 for proposals aimed at improving thermostability of a variety of new vaccine platforms, so as to improve equitable access.
Representing a significant milestone for Vaxxas, it builds on its pre-clinical portfolio into mRNA vaccines, with the potential to expand the company’s vaccine technology pipeline across a number of diseases, including COVID-19.
Vaxxas’ proprietary high-density microarray patch (HD-MAP) technology is currently being evaluated in a phase 1 clinical study of the first needle-free COVID-19 vaccine, having demonstrated successful delivery in a preclinical study, published as a preprint publication.
The HD-MAP utilises an ultra-high-density array of projections – which are invisible to the naked human eye – applied to the skin to rapidly deliver a vaccine to immune cells immediately below the skin’s surface.
Furthermore, its dry-coating technology could eliminate or significantly reduce the need for vaccine refrigeration during storage and transportation: Vaxxas’ HD-MAP vaccines have been shown to remain stable for up to a year at 40°C (104°F).
This also opens the possibility for mass vaccinations that could be self-administered via patch at home, thereby addressing issues of equitability. For instance, HD-MAP patches could be posted not only to people’s homes, but to workplaces and schools, circumventing delays and the waste and inconvenience of traditional needle-and-syringe vaccine scheduling and administration.
Under the terms of the funding agreement, Vaxxas commits to achieving equitable access to the outputs of the project, including prioritisation of supply for low-income and middle-income countries (LMICs), as well as production of vaccine volumes required to meet public health needs, with affordable pricing, and the potential technology transfer to LMIC manufacturers in line with CEPI’s Equitable Access Policy.
Chair of CEPI, Jane Halton, said: “As we have witnessed with COVID-19, equitable access to vaccines must be at the heart of any effective pandemic response […] Combining Vaxxas’ vaccine-patch technology with the speed and effectiveness of mRNA vaccines could produce a tool that is not only suited as a rapid-response platform for use against unknown pathogens, but could also serve as an additional means to get life-saving vaccines to the most vulnerable populations around the world.”
CEO of Vaxxas, David Hooey, said: “Earning this significant funding from one of the world leaders in vaccine development is a great honour, and validates the benefits offered by Vaxxas’ HD-MAP platform in the fight against global epidemic and pandemic threats.”
Before COVID-19, CEPI’s work focused on developing vaccines against Ebola virus, Lassa virus, Middle East Respiratory Syndrome coronavirus, Nipah virus, Rift Valley Fever virus, and Chikungunya virus.
In February 2022, the UK said it would pledge £160 million to CEPI to speed up vaccine development. The government has supported the Coalition since its inception and had previously provided £276 million for the organisation’s life-saving work since 2018.