Patients call for end to two-year row over CF drug funding
UK patients are stepping up efforts to resolve the “heartbreaking” row that is denying their families access to Vertex’s groundbreaking cystic fibrosis (CF) drug Orkambi, after Sweden became the latest European country to reimburse it.
Vertex has reached an agreement with the Swedish health system to reimburse Orkambi, the first medicine to treat the underlying cause of cystic fibrosis in people with two copies of the F508del mutation, aged six or over.
Reimbursement will begin from July 1, and the access agreement provides a framework for the assessment and access of future CF drugs, Vertex said.
Sweden will be added to a growing list of other EU countries funding Orkambi (Iumacaftor/ivacaftor) for cystic fibrosis – Austria, Germany, Ireland, Italy, the Netherlands and the US are among the countries where the drug is available.
But this is not the case in the UK, where Orkambi has been rejected for regular NHS funding in England by NICE and by the Scottish Medicines Consortium (SMC).
Vertex is still in talks with NHS England over funding for Orkambi after NICE said it was too expensive in 2016, but patient groups are calling for progress.
At the end of the week, patients and their families will march outside the Department of Health, the offices of Vertex, and Belfast City Hall, calling for the drug to be reimbursed.
They will present a floral tribute to an estimated 200 people who have died from the disease since the funding row began.
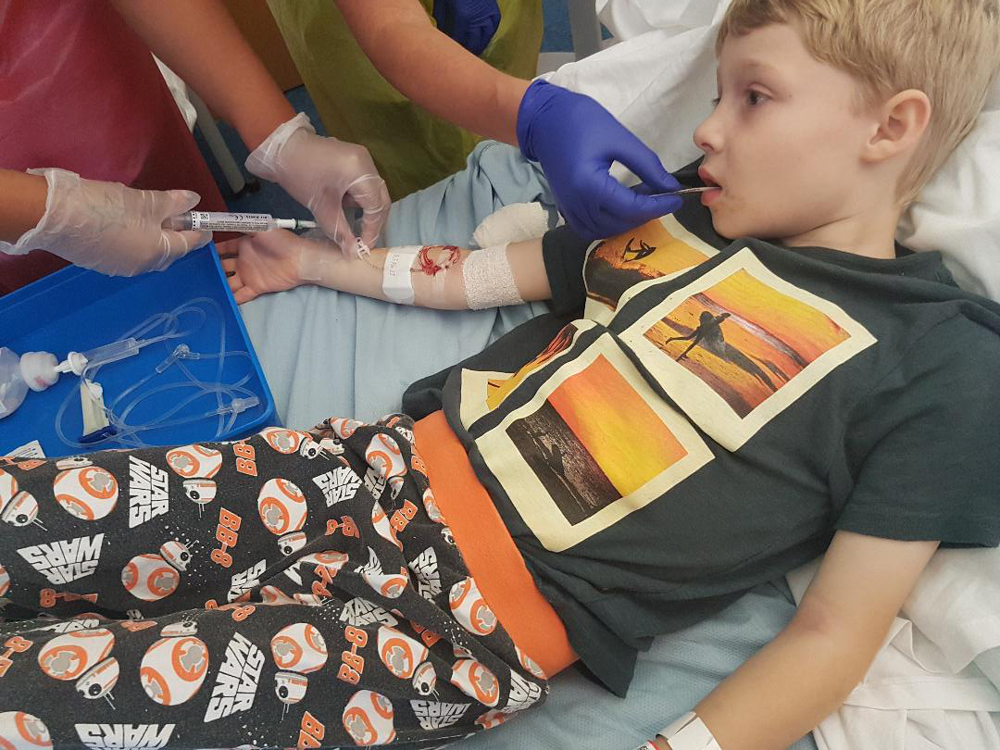
Christina Walker, from the patient group ukneedsorkambi, said her family is “desperate” for Orkambi to be made available, as her eight year-old son Luis (pictured above) has the disease and is eligible for treatment.
Walker said Orkambi could reduce the risks of infection and conditions such as CF-related diabetes.
She told pharmaphorum: “It is heartbreaking and frustrating. There is this thing that is just out of reach that we are hearing could make a real difference.”

Christina Walker and Luis
Walker argues that the situation is particularly unfair as Vertex's Kalydeco (ivacaftor), which only works in a small group of patients, has been funded because it was assessed using NICE's pathway for ultra-rare diseases.
"Kalydeco patients now enjoy a near-normal life because they are 'lucky enough' to have a rare type CF, so the NHS will fund their drug due to small patient group numbers," said Walker, who urged Vertex to cut its "excessively high" prices.
Vertex is asking for a deal covering reimbursement for its entire CF drug portfolio and any drugs approved in the future, and is keen for progress as with 10,000 people with CF as the UK has the second largest patient population in the world.
Another meeting between Vertex and NHS England is scheduled for the end of the month – and Walker urged the two parties to come to an agreement following similar deals in Ireland and the Netherlands.
“It is a wonderful opportunity to deal with something that is costly in terms of hospital beds, and for the patients whose lives are limited,” said Walker.
A Vertex spokesperson said: “We will continue to be creative, bringing solutions that are consistent with multiple agreements we’ve reached in other countries across Europe. In order to secure an agreement, we urge NHS England and NICE to be flexible partners in these discussions as we work together toward an agreement as quickly as possible in this exceptional circumstance.”
CF is a rare disease causing continuous damage to multiple organs from birth, and a build-up of sticky mucus in the lungs causes progressive and permanent damage, severe infections, and premature death.
NHS England declined to comment while discussions are ongoing.












