Beyond obesity: How GLP-1s are changing the game for hundreds of diseases
The introduction of GLP-1 treatments for obesity has, in a short space of time, completely upended the clinical research industry. Recent data shows that obesity is now the sixth most-studied disease in the world, thanks to the rise of this transformative new class of medicines.
But obesity isn’t the only condition benefiting from the GLP-1 boom. Recent analysis from Phesi has found that more than 100 diseases are now being investigated in the context of GLP-1 usage. The data shows a clear trajectory – GLP-1s began in diabetes, moved into obesity, and we are now seeing increasing application across a spectrum of diseases. The key to understanding this trend lies in researchers’ growing interest in disease clusters with shared risk profiles, and how GLP-1s could interact with multiple disease pathways.
The study
The global analysis looked at real world data (RWD) from 583 recruiting clinical trials or trials about to start involving GLP-1 receptor antagonists. This included trials directly evaluating GLP-1s as interventions, as well as those exploring comorbidities and modulatory effects. The study analysed 1.8m patients about to be treated by GLP-1 inhibitors from 69,652 hospitals and clinics in 81 countries over the past 20 years. This ensured a precise understanding of key characteristics specific to the indication and patient population under study, including age, sex, comorbidities, outcome measures, and concomitant medications.
Although obesity and diabetes are still the most-studied diseases for GLP-1 treatment, the analysis shows that more than 100 diseases are being studied using GLP-1 therapies, including polycystic ovary syndrome, hypoglycaemia, and chronic kidney disease (see Table 1).
| Indication | Studies | Indication | Studies |
| Obesity | 63 | Chronic Kidney Disease | 7 |
| T2DM | 33 | Endometrial Cancer | 6 |
| T1DM | 18 | Cardiovascular Diseases | 6 |
| MASLD | 15 | Alcohol Use Disorder | 6 |
| Polycystic Ovary Syndrome | 12 | Glucose Metabolism Disorders | 5 |
| Prediabetes | 11 | Pancreatic Cancer | 5 |
| Hypoglycaemia | 9 | Crohn’s Disease | 4 |
| Breast Cancer | 4 | Bariatric Surgery Candidate | 4 |
| Cystic Fibrosis | 4 | Binge Eating Disorder | 4 |
| Heart Failure | 4 | Prostate Cancer | 3 |
| Metabolic Syndrome | 3 | Insulin Resistance | 3 |
| Steroid-Induced Diabetes | 3 | Gestational Diabetes Mellitus | 3 |
Table 1: Top 26 indications where GLP-1 receptor antagonists are being studied
Towards disease clusters
Why is interest growing in GLP-1s as a broader modulatory pathway across indications? Many of these conditions have comorbidities with weight and metabolic diseases. It’s therefore not surprising that GLP-1s have an impact on hundreds of other conditions. As a result of this crossover, scientific curiosity is increasingly centred on understanding how weight and metabolic factors intersect with other disease areas, including cardiovascular, inflammatory, and neurological conditions.
Osteoarthritis is one example: almost 80% of osteoarthritis patients are overweight or obese. Excess weight can apply extra pressure on joints such as the knee. Reducing weight could have a positive impact on the disease, either in terms of prevention or pain reduction in existing patients. Clinicians shouldn’t restrict themselves to simply giving painkillers to reduce symptoms; the issue could be addressed in a more rounded way. For example, the combination of a drug to combat arthritis and a reduction of weight may have a synergistic impact.
Metabolic dysfunction-associated steatohepatitis (MASH) is another condition that demonstrates the significance of disease clusters. Phesi analysis finds that MASH comorbidities include metabolic syndrome (42% of patients) and type 2 diabetes (28%); both can be treated with GLP-1s. Meanwhile, 42% of MASH patients are also obese. As a result, 12% of MASH patients are already taking GLP-1s. There is therefore potential to investigate whether adequate GLP-1 therapy may delay MASH onset. Further analysis has found that MASH patients in the US are younger (mean age 49.5 yrs vs 53.4 yrs), heavier (mean BMI 31.2 kg/m² vs 25.9 kg/m²), and sicker (higher comorbidities) than their Chinese counterparts. It is highly likely that the experimental intervention outcomes would be different between the two patient populations.
Increasingly, the goal for the industry is not just treating obesity, but treating the entire constellation of conditions associated with it. Step by step, scientists are realising that they no longer need to limit themselves to studying isolated diseases. Human diseases often coexist – a patient may develop hypertension, then diabetes, and later cancer, alongside many other conditions in between.
To be in a better position to address these disease clusters, researchers need to define and quantify the exact relationship between obesity and other diseases. Understanding a typical patient in detail via a digital patient profile is vital. That way, researchers can look at issues such as comorbidities and the severity of the disease to find the best way to help these patients.
The industry is still at the early stages of understanding disease convergence, and hypotheses are still emerging that will require significant real-world data (RWD) to substantiate. The good news is that as GLP-1 usage grows, the volume of real-world data also increases rapidly. Sophisticated clinical data analytics combining RWD and disease modelling will unlock insights that optimise clinical operations, reduce costs, drive down patient and investigator site burden, and enhance regulatory strategies. These insights will also afford sponsors and clinicians a deeper understanding of longitudinal outcomes in treated patients and provide data that can be fed back into analysis.
Patient-centric clinical data science, propelled by artificial intelligence, not only helps us to better define the treatment context known to us, but also to explore innovative treatment contexts with unprecedented precision.
Now that we have better knowledge, there’s no reason not to leverage it to treat patients more effectively. GLP-1s exemplify the shift from siloed disease treatment to a holistic, patient-centred approach, where a single intervention could address multiple related conditions.
Growing pains for GLP-1 trials
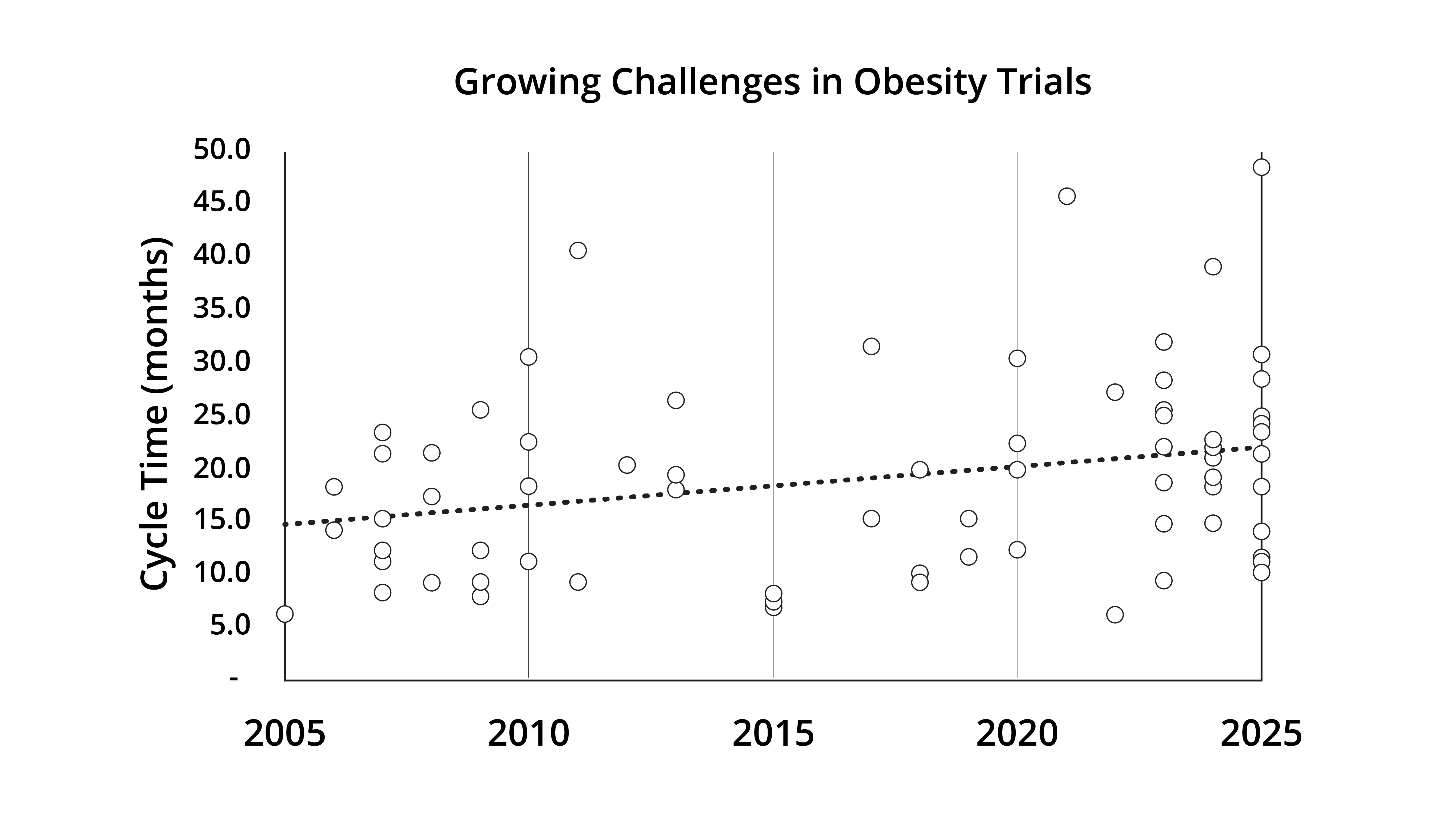
But as GLP-1 trials become more commonplace, there are also some knock-on effects that the industry needs to navigate. Notably, cycle times for these trials are increasing: the average duration of obesity trials has grown over the past two decades, with trials that used to take 10–20 months now often taking 25-45 months (see Fig.1).
Fig.1. Trend in clinical trial cycle times for obesity-related studies between 2005 and 2025
When an effective treatment exists, patients are less willing to participate in trials testing something even newer. Similarly, the medical community may feel less urgency than before. This dynamic isn’t unique to obesity; it applies across many diseases.
Meanwhile, clinical specialisms will be forced to adapt to this new paradigm. As diseases like cardiovascular conditions, MASH, and diabetes increasingly converge, physicians and trial designers must move beyond single-disease expertise to multi-indication decision-making. Operationally and to support this shift, clinical development organisations need to become smarter and more data-driven to ensure portfolios and programmes remain viable against this evolving backdrop.
In one sense, these are welcome challenges. For decades, the industry has made enormous efforts to manage obesity globally. Now that we have something that works extremely well, clinical development becomes more challenging. GLP-1s are forcing a thorough re-evaluation of how we define, diagnose, and treat disease – and are doing so at unprecedented speed. COVID was the last great disruptor for the clinical trial industry, but the sector needs to be prepared for the next disruptor. With obesity rising to become the sixth most-studied disease in the world, and likely to continue seeing an increase in clinical activity, that disruptor could very well be GLP-1s.
It is extremely encouraging to see so much rapid progress in this area. GLP-1s exemplify the idea that prevention, viewed through the lens of disease clusters, is emerging as the new blockbuster. And with the right data in hand, sponsors can target the right patients earlier, saving time, money, and lives.
About the author
Dr Gen Li, founder and CEO, Phesi
Dr Gen Li founded Phesi in 2007 with the aim of revolutionising the clinical trials industry. Prior to founding Phesi in 2007, Dr Li was head of productivity for Pfizer Worldwide Clinical Development, a position he assumed following Pfizer’s acquisition of Pharmacia, where he delivered the first implementation of productivity measurement for clinical development.
Previously, he earned his PhD in Biochemistry from Beijing University, and an MBA from the Johnson Graduate School of Management at Cornell University.
Supercharge your pharma insights: Sign up to pharmaphorum's newsletter for daily updates, weekly roundups, and in-depth analysis across all industry sectors.
Click on either of the images below for more articles from this edition of Deep Dive: GLP-1 2026