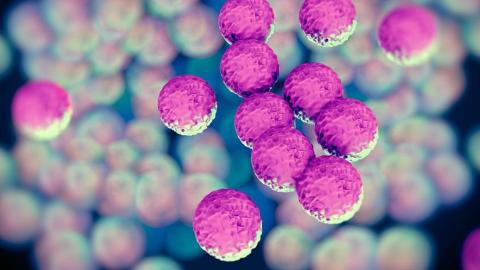
Basilea completes ceftobiprole marathon with FDA approval
… Zevtera or Mabelio in Europe, China, and Canada – as well as in … to provide an update on the partnership “around mid-year.” …
Newsletters and Deep Dive digital magazine

… Zevtera or Mabelio in Europe, China, and Canada – as well as in … to provide an update on the partnership “around mid-year.” …

… a timely and efficient manner. Partnership between the central … and the growing R&D efforts in China, an industry-wide need to …
… and glucagon, developed in partnership with Zealand Pharma, … worth up to $2 billion with China’s Suzhou Ribo Life Science …

… day after Roche pulled out a partnership on its underperforming … markets, except for Greater China (mainland China, Hong Kong, …

… out of a $1.3 billion R&D partnership with I-Mab on … in clinical development in China. Photo by Nadine Shaabana on …

… by Ally Bridge Group. US and China-based ProfoundBio will use … recently signed a wide-ranging partnership with Pfizer’s ADC unit …

… Researchers in the UK and China have used proteomics data … Challenge project in the UK in partnership with Alzheimer’s …
… approved in the US, Japan, and China, and under regulatory review … Challenge project in the UK in partnership with Alzheimer’s …

… multi-target collaboration with China’s Suzhou Ribo Life Science … at Boehringer. “This new partnership is part of our …

… recently pulled out of an R&D partnership with I-Mab on … the drug independently in China. BMS, meanwhile, switched …

… GSK has turned to China’s Hansoh Pharma to add … include MSD’s $22 billion partnership with three candidates …

… to a HER3-directed ADC from China’s SystImmune in a deal that … after a smaller $180 million partnership with Orum Therapeutics …

It’s no secret that the UK life sciences sector is facing skills shortages – in a 2020 report, the Science Industry Partnership’s Life Sciences 2030 Skills Strategy predicted that 133,000 e


Explore “Innovation as Strategy” framework, providing a guide for employing innovative strategies in life sciences.

In this edition of Deep Dive, Unpacking complex choreography in the biologics patent dance, Alexion examines the urgent need for accelerated access programmes for rare disease treatments, and Putna

It will come as no surprise to anyone in the healthcare space that doctors today are under an inordinate amount of pressure.

We are living in an era of unprecedented advances in cancer treatments with transformative therapies being regularly approved.

The 2nd mRNA Quality Control & Compliance Summit 2024 returns this June 25-27 (Boston, MA), as the sole industry-centred forum dedicated to showcasing technical data and though

The commercial success of cell therapies is continuing to grow!