The hidden access gap in Europe’s lung cancer hospital data

Ask a pharma commercial team where lung cancer patients are treated in France and most will point to major academic centres such as Gustave Roussy, Institut Curie, or the Assistance Publique network in Paris. These institutions are influential and highly visible. But they represent only a fraction of where patients actually receive treatment.
New hospital-level analysis shows a provider landscape that is far more concentrated, regionally uneven, and strategically complex than most commercial plans assume.
Using diagnostic discharge data for malignant neoplasm of bronchus and lung (ICD-10 C34), sourced from ATIH (l’Agence Technique de l’Information sur l’Hospitalisation) and linked with PurpleXtra’s own hospital database, we mapped every hospital in France treating lung cancer. The findings challenge a persistent industry assumption: that national markets can be approached through broadly uniform commercial coverage.
In the most recent reporting year, 578 hospitals treated 55,976 lung cancer cases. Treatment activity, however, is sharply concentrated.
The 20% that treat 70%
Ranking hospitals by annual case volume reveals a clear Pareto distribution. Just 116 hospitals, 20.1% of treating facilities, account for 70% of all lung cancer cases. Expanding to the top 245 hospitals captures 90% of national volume.
The remaining 333 hospitals share just 10% of cases between them, many treating fewer than 20 patients annually.
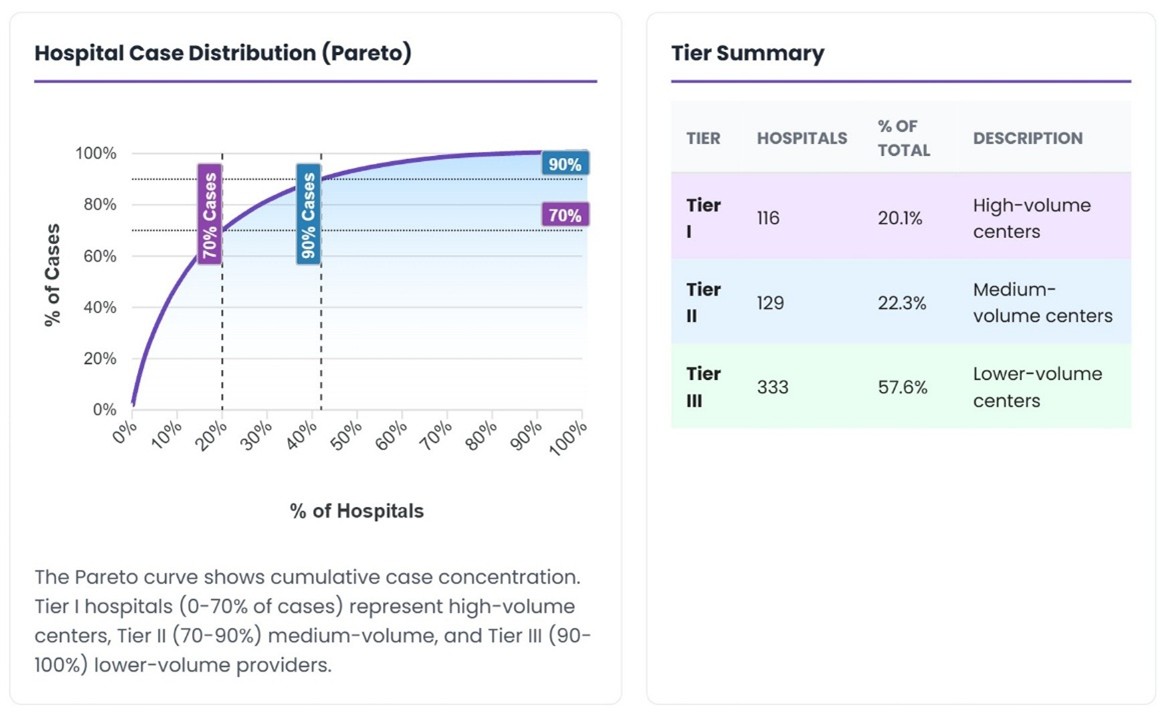
Figure 1: Hospital case distribution (Pareto) showing cumulative concentration across tiers.
For commercial teams, the implication is immediate. A visit to a hospital managing 400 cases annually operates in a fundamentally different value equation from one treating 15. Yet, deployment models are still frequently structured around geography or specialist headcount, rather than treated patient volume.
The result is a structural mismatch between commercial effort and where patients are actually treated.
Not one market, but thirteen
The national picture already challenges assumptions. At regional level, the strategic implications become clearer still.
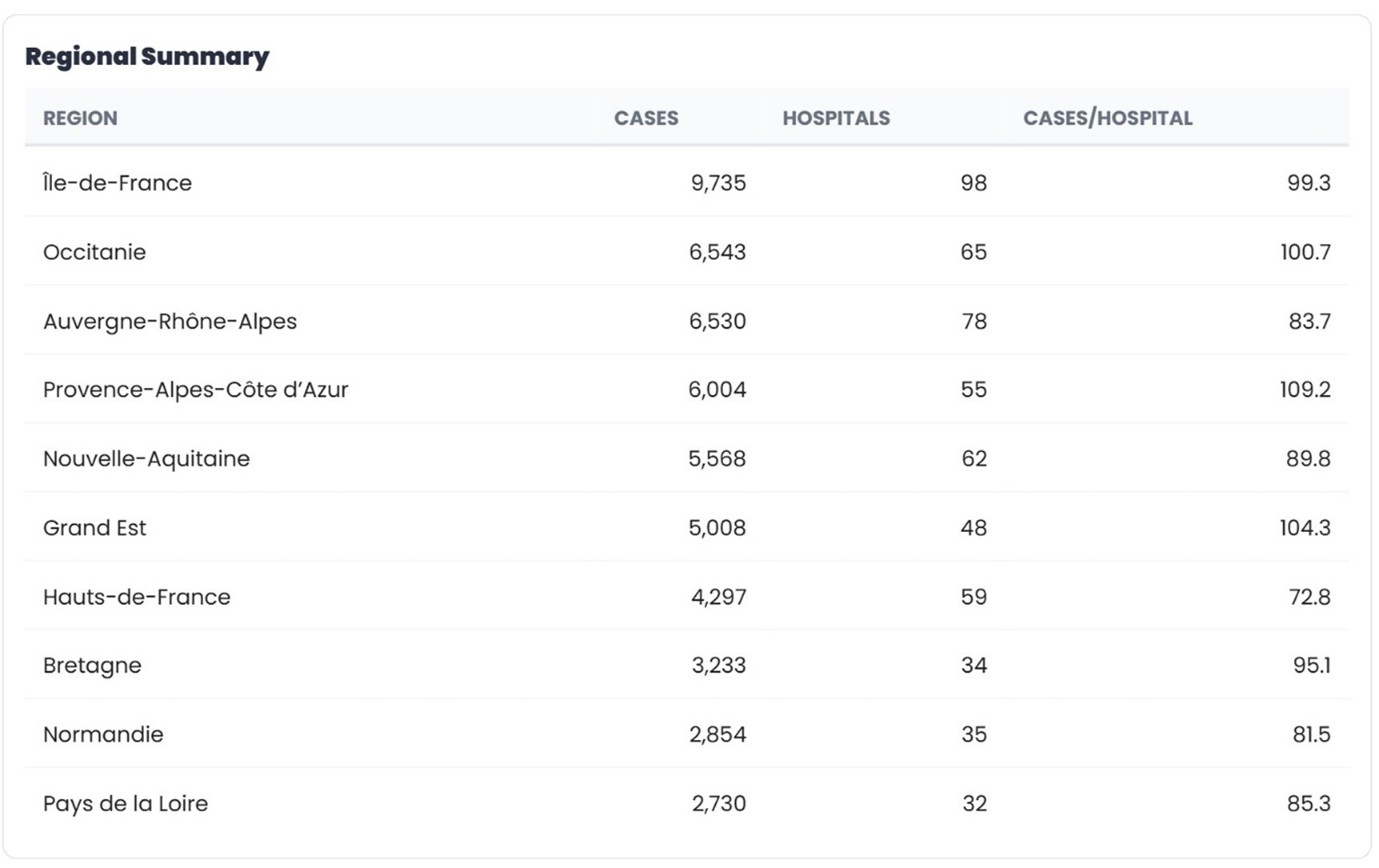
Île-de-France leads with 9,735 cases across 98 hospitals, averaging 99.3 cases per site. Provence-Alpes-Côte d’Azur shows similar concentration, allowing relatively small field teams to reach a large share of patients.
By contrast, Hauts-de-France treats 4,297 cases across 59 hospitals, averaging just 72.8 cases per hospital. Auvergne-Rhône-Alpes records the second-highest national volume, but distributes activity across 78 hospitals, reducing average site volume to 83.7.
Identical commercial effort therefore produces very different patient reach depending on regional provider structure.
This variation raises a strategic question pharma teams rarely address explicitly: should resources concentrate where patients cluster, or extend into dispersed regions where lower-volume hospitals may welcome greater clinical engagement?
Concentrate or cover?
The argument for concentration is straightforward. High-volume hospitals influence formularies, host key opinion leaders, and deliver the greatest return on each interaction. Securing strong positioning in the 116 hospitals treating 70% of patients represents the fastest route to market share.
But concentration has limits. The 333 Tier 3 hospitals collectively treat more than 5,600 patients annually, often with fewer treatment options and less access to clinical trials. For companies positioning around access and equity, there is both a moral and commercial case for engagement beyond major centres.
The practical solution is differentiated engagement. Tier 1 centres warrant dedicated account management and sustained medical engagement. Tier 2 hospitals suit hybrid models combining in-person interaction with digital channels. The long tail of Tier 3 hospitals is most efficiently supported through regional education programmes and scalable digital outreach.
What makes this approach newly achievable is data precision. Hospital-level treated patient volumes now allow targeting based on actual care delivery, rather than institutional reputation or proxy metrics such as bed count.
Where research activity concentrates
Clinical trial activity adds another layer to the concentration picture. France hosts 851 ongoing lung cancer trials, with 154 initiated in 2025 alone, heavily clustered within a small group of centres.
Gustave Roussy leads with 236 active trials, followed by Centre Léon Bérard in Lyon and Institut Curie in Paris.
For pre-launch planning, this creates a dual targeting dynamic. Hospitals conducting the most research are often, but not always, those treating the most patients. Where treatment volume and research intensity overlap, the case for sustained medical engagement is clear. Where they diverge, high-volume but research-light centres may represent overlooked partnership opportunities.
The European dimension
France reflects a broader European pattern. Across 15 countries, 604,622 lung cancer cases are treated in 3,781 hospitals. Treatment intensity ranges from 275 cases per 100,000 population in the Netherlands to just 15 in Ireland, with France sitting in the lower half of the range.
Provider structure varies just as dramatically. Switzerland has 2.3 hospitals per 100,000 population treating lung cancer; the Netherlands has 0.6. Commercial models that work efficiently in one country rarely transfer directly to another.
For multinational teams, country strategies must be built from provider-level realities, rather than scaled from incidence estimates or regional averages.
From data to deployment
The concentration patterns observed in France are not unique to lung cancer. Across indications and countries, healthcare delivery consistently follows a Pareto structure, with a minority of hospitals treating the majority of patients.
What has changed is visibility.
Provider-level data now makes it possible to align commercial deployment with where patients are actually treated. Geography-based coverage models and reputation-driven targeting are increasingly difficult to justify when precise treatment data is available.
The access gap is measurable. The data to close it already exists. Competitive advantage will belong to organisations that move fastest from assumption-based planning to evidence-based deployment.
NB: Hospital discharge activity is sourced via ATIH (l’Agence Technique de l’Information sur l’Hospitalisation). Clinical trial data is from ClinicalTrials.gov.
About the author

Bob Tulloch is co-founder of PurpleXtra Limited, a healthcare data company mapping hospital-level treatment activity across 15 European countries using Freedom of Information-sourced diagnostic discharge data.











