Kite begins EU trial of CAR-T therapy
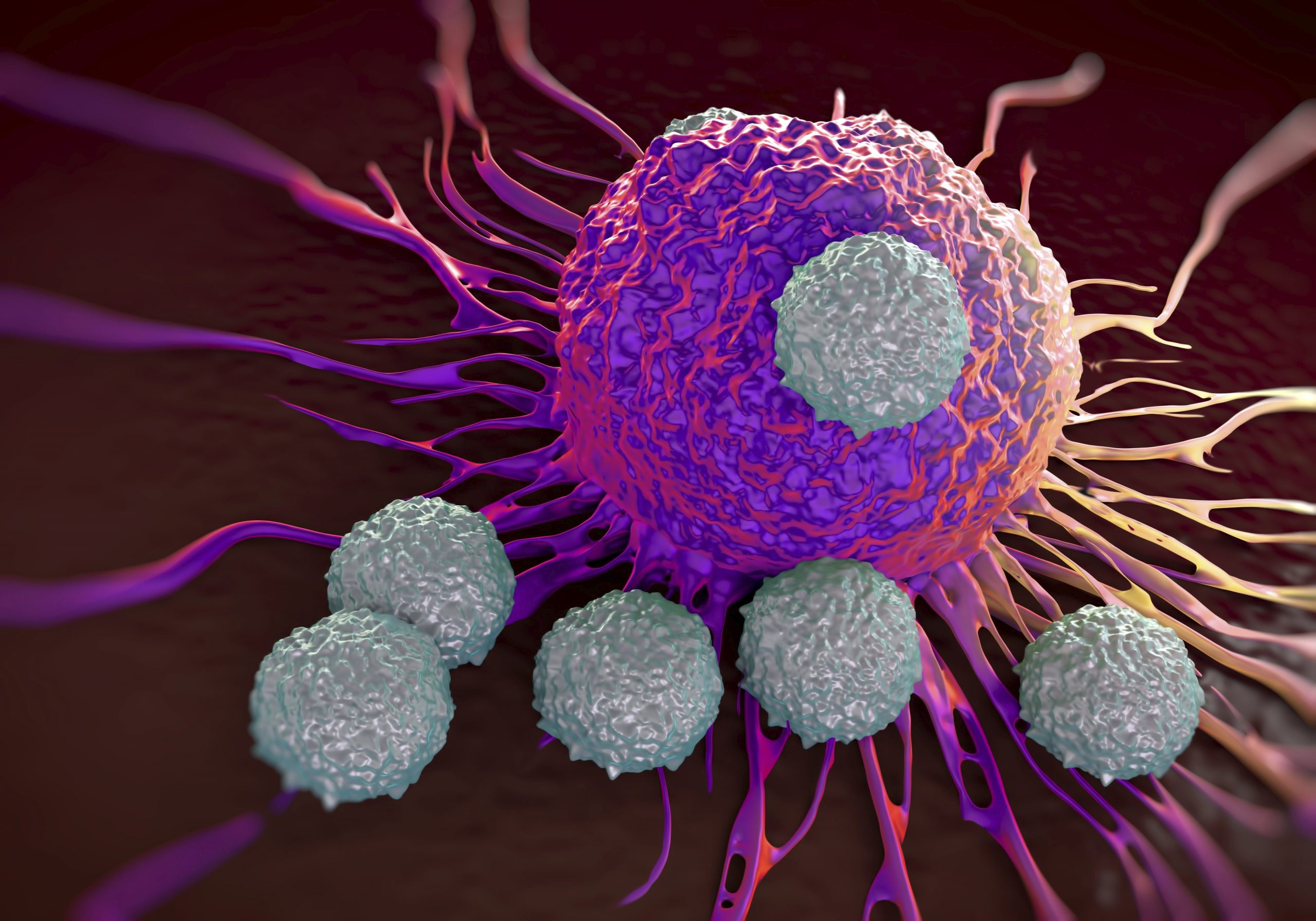
The first European patients are set to receive Kite Pharma’s CAR-T cancer therapy after the US biotech started EU trials.
The first patient to receive the treatment known as axicabtagene ciloleucel in the expansion of the ZUMA-1 trial was treated at the Academic Medical Center (AMC) in Amsterdam.
Patients involved in the trial will have relapsed/refractory diffuse large B-cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma (PMBCL), and transformed follicular lymphoma (TFL), all forms of aggressive non-Hodgkin lymphoma.
The EU trial follows the US-based cohort of the ZUMA-1 trial, which produced strong results in this heavily treated population, who had previously faced a dismal outlook.
In ZUMA-1, 82% of patients achieved a response after a single infusion of axi-cel. A further 44% remain in ongoing response, including 39% in complete response at a median follow-up of 8.7 months.
Kite has been granted access to Priority Medicines (PRIME) regulatory support in the EU for the treatment of refractory DLBCL. Access to the PRIME initiative is granted by the European Medicines Agency to support the development and accelerate the review of new therapies to treat patients with a high unmet need.
Axicabtagene ciloleucel is an investigational therapy in which a patient's T cells are engineered to express a chimeric antigen receptor (CAR) to target the antigen CD19 - a protein expressed on the cell surface of B-cell lymphomas and leukaemias - and redirect the T cells to kill cancer cells.
While Novartis is ahead of Kite in the US in the race to get a CAR-T therapy to market, with an FDA decision due in the coming weeks, Kite has taken the lead in Europe by filing its rival with the European Medicines Agency first.
If all goes well the treatment could be on the market in Europe next year. Professor Marie Jose Kersten, principal investigator and head of the Department Hematology at Amsterdam’s AMC, said: “As a lymphoma specialist, I am gratified that we can now offer this potentially transformative therapy to patients with refractory, aggressive NHL who previously had no other therapeutic options.”











