The expanding role of early-phase trials in modern drug development
Early-phase clinical trials are undergoing a quiet but consequential transformation. Once considered a relatively narrow checkpoint focused on basic safety and tolerability, these early-phase studies are now also expected to provide insights that can influence how efficiently programmes progress and how challenges are addressed later in development.
Within a rapidly changing research and development landscape, multiple factors are driving this change. Scientific innovation is producing therapies with more complex mechanisms and less predictable translation from preclinical models. Regulators are seeking clearer justification earlier in development, particularly for novel modalities. Investors and internal governance teams are scrutinising early data more closely, using these results to inform funding and portfolio decisions long before pivotal trials begin.
Together, they have moved early-phase development from a preparatory exercise to a strategic foundation. What sponsors decide during early planning, including how studies are designed, where they are conducted, and how capabilities are aligned, can significantly strengthen or weaken later-phase execution.
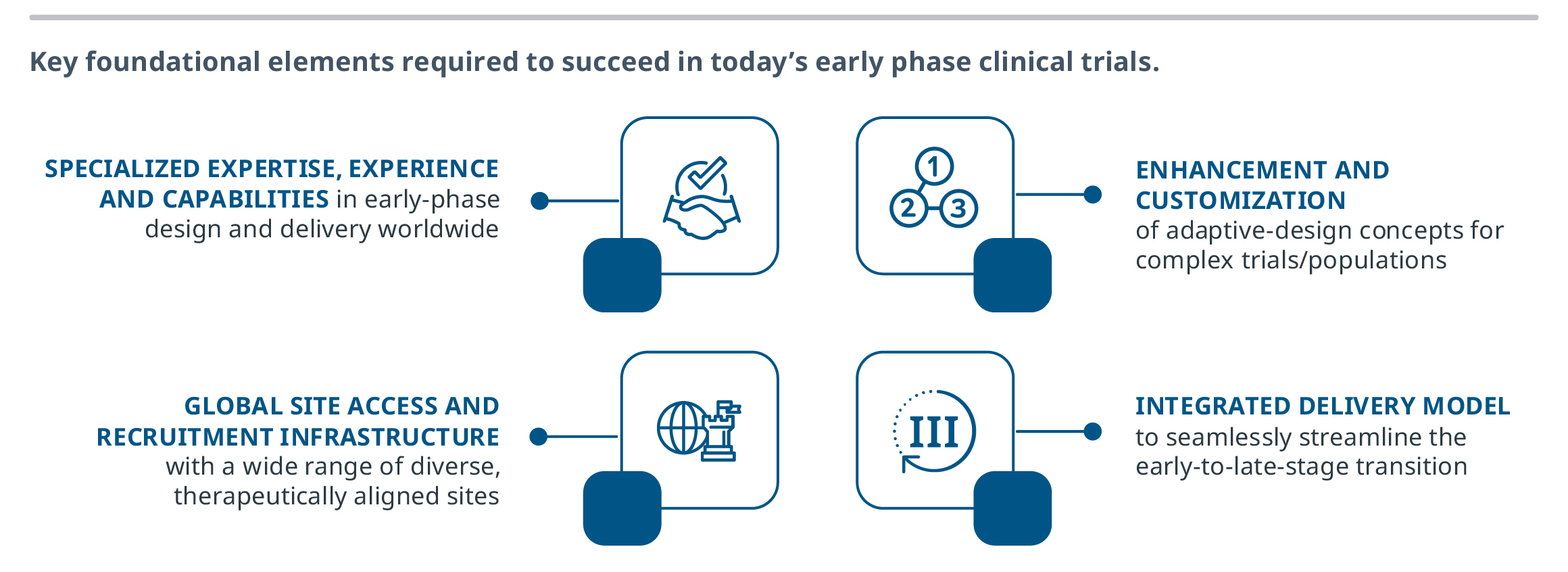
Four interdependent considerations now require intentional foresight to ensure early-phase studies support long-term development objectives, rather than limit flexibility in later phases when needed most.
Translational complexity demands depth of early-phase expertise
Modern early-phase trials ask far more of clinical teams than they did even a decade ago. Sponsors are working with increasingly sophisticated therapies, many of which challenge traditional assumptions about dose selection, safety margins, and pharmacologic behaviour in humans. Given this context, early development can no longer rely on standardised approaches or generalised experience.
The depth and breadth of expertise applied at the outset can help ensure that early-phase studies generate insights that guide next steps, rather than data that offers limited strategic value. Moving from preclinical data into first-in-human studies, especially when therapies are not as predictable to translate from experimental models to patients, depends on close coordination across clinical, regulatory, and therapeutic experts. Each discipline contributes a perspective that helps refine dosing strategies, anticipate safety considerations, and align study objectives with downstream needs.
From a holistic view, these early decisions set expectations that persist across development phases. Initial dose levels, escalation rules, and cohort structures inform early outcomes, as well as how regulators and investigators interpret data in later phases. Programmes that do not fully account for these design considerations early on may need to revisit core assumptions later in development, often with meaningful cost and timing implications.
By contrast, early-phase programmes supported by specialised, cross-functional expertise are better positioned to balance safety with efficiency and to ensure that the data generated provides a reliable foundation for later-stage decision-making.
Designing for evidence-based adaptation
As expectations for early-phase studies increase, study designs must evolve accordingly. Clinical trial sponsors are increasingly looking to early-phase studies to deliver more than isolated safety readouts, using them to support confident advancement decisions while minimising unnecessary risk.
This evolution has driven increased adoption of adaptive, data-informed trial designs that enable protocol modifications in response to emerging evidence. When rigorously planned and clearly defined before a study begins, such approaches can enhance the efficiency of evidence generation, minimise unnecessary participant exposure, and potentially shorten development timelines. Importantly, their success is contingent on intentional and methodologically sound design decisions made early in development.
Designs that are reactive, rather than anticipatory, often struggle to keep pace with emerging insights. Mid-study amendments, delayed adaptations, or misaligned endpoints can undermine both efficiency and interpretability. Early planning is therefore essential to ensure that study designs anticipate the need for change by defining escalation pathways, cohort transitions, and assessment schedules upfront to support evolving development questions.
Equally important is acknowledging that flexibility is essential in early-phase trials, but that it requires careful management given the operational and analytical complexity it may introduce. Having rapid data review, cross-functional coordination, and clear governance processes in place can help sponsors act on emerging signals in real time. When these elements are aligned early, designs support learning and momentum for development. Without that alignment, flexibility can complicate execution, rather than support it.
Getting enrolment right earlier: Strategic choices shaping development
Patient enrolment is a long-standing operational challenge, but in today’s early-phase development environment, it is increasingly a strategic consideration, requiring careful and ongoing attention. The ability to enrol appropriate participants, both quickly and responsibly, directly influences data quality, study timelines, and downstream credibility.
Early-phase trials often face distinct recruitment hurdles. The participant benefit may be uncertain, safety profiles are still emerging, and participation can place significant demands on them. Investigators may also be cautious without strong scientific rationale and confidence in trial oversight.
These realities make site selection and recruitment planning foundational decisions, rather than logistical afterthoughts. Sponsors must consider not only timelines, but also therapeutic alignment, access to patient populations, and long-term scalability. Sites that are well matched to a study’s scientific requirements and supported by robust recruitment infrastructure are more likely to deliver consistent, high-quality data.
Geographic strategy also plays a growing role in early-phase development. Access to various populations, specialised capabilities, and region-specific efficiencies can enhance enrolment and regulatory positioning later. Early choices about where and with whom studies are conducted often determine whether programmes can expand smoothly or face bottlenecks when moving into later phases.
Viewing participant access through a strategic lens can help sponsors mitigate one of the more frequent causes of downstream delays and uncertainty.
Reducing friction at key development transitions
While early-phase data now receives greater scrutiny, development efforts can remain fragmented. Early-phase studies may be conducted without fully accounting for how teams, data, and insights will be carried into subsequent phases, with implications that emerge later in development.
Fragmentation introduces subtle but meaningful risk. Institutional knowledge may be lost, forcing teams to reinterpret earlier decisions without full context. Differences in data standards and operational processes can complicate integration, while onboarding delays slow momentum at critical transitions.
Early-phase planning that incorporates a longer-term or holistic view of development can mitigate these risks. Continuity of scientific leadership, alignment of data frameworks, and early consideration of how studies will scale all contribute to a smoother progression when advancement decisions are made.
However, it is important to note that integration does not require rigidity. More than ever, staying fluid will be critical, ensuring early insights remain accessible and actionable as development evolves. Programmes that maintain this continuity are better positioned to build upon early momentum instead of rebuilding it at each stage.
Foresight early on: Paying it forward
The evolving role of early-phase clinical trials reflects a broader truth: decisions made early carry increasing weight in shaping clinical success. Sponsors who recognise this shift are adjusting how studies are planned and conducted, not to add complexity, but to better support development progress.
By investing early in specialised expertise, structuring study design with intent, strategic access to participants, and development continuity, sponsors can generate evidence that supports confident advancement while minimising downstream friction. These considerations do not eliminate uncertainty, but they can help to ensure that uncertainty is addressed early when adjustments are still feasible.
With development timelines tightening and evidentiary standards increasing, early-phase clinical development has become a defining decision point. How studies are designed and executed early on now plays a central role in determining whether promising therapies advance efficiently or encounter constraints that limit their potential later.
About the authors
Elizabeth (Liz) Allen, PhD, is vice president of the Early Phase Development Centre of Excellence, Scientific Strategy, at IQVIA. She provides scientific leadership and expert advice on the design and conduct of early-phase clinical studies, from first-in-human through proof of concept.
With more than 35 years of experience in clinical pharmacology, Allen has deep expertise in complex adaptive study design and regulatory strategy, shaping rigorous, decision-focused early development programmes.
Raymond Cook, MD, is vice president of Medical Strategy for Early Phase Development at IQVIA. He is responsible for oversite of the Early Phase Center of Excellence, which provides early phase clinical design and strategy to support clinical trial sponsors in the development and implementation of their early clinical development plan.
With two decades of combined clinical, academic, and industry experience, Cook brings deep expertise in protocol development, regulatory submissions and cross-functional clinical governance across early-phase development programmes.
About IQVIA
IQVIA (NYSE:IQV) is a leading global provider of clinical research services, commercial insights and healthcare intelligence to the life sciences and healthcare industries. IQVIA’s portfolio of solutions are powered by IQVIA Connected Intelligence to deliver actionable insights and services built on high-quality health data, Healthcare-grade AI, advanced analytics, the latest technologies and extensive domain expertise. IQVIA is committed to using AI responsibly, with AI-powered capabilities built on best-in-class approaches to privacy, regulatory compliance and patient safety, and delivering AI to the high standards of trust, scalability and precision demanded by the industry. With approximately 88,000 employees in over 100 countries, including experts in healthcare, life sciences, data science, technology and operational excellence, IQVIA is dedicated to accelerating the development and commercialisation of innovative medical treatments to help improve patient outcomes and population health worldwide.
IQVIA is a global leader in protecting individual patient privacy. The company uses a wide variety of privacy-enhancing technologies and safeguards to protect individual privacy while generating and analysing information on a scale that helps healthcare stakeholders identify disease patterns and correlate with the precise treatment path and therapy needed for better outcomes. IQVIA’s insights and execution capabilities help biotech, medical device and pharmaceutical companies, medical researchers, government agencies, payers and other healthcare stakeholders tap into a deeper understanding of diseases, human behaviours and scientific advances, in an effort to advance their path toward cures. To learn more, visit www.iqvia.com.
Follow us on LinkedIn
Supercharge your pharma insights: Sign up to pharmaphorum's newsletter for daily updates, weekly roundups, and in-depth analysis across all industry sectors.
Click on either of the images below for more articles from this edition of Deep Dive: Market Access 2026