Why PM 1.0 will not create radical healthcare change prompting the need for PM 2.0

Peter Keeling explores the concept of personalised medicine 2.0 in our personalised medicine themed month.
The six forces behind 2.0 anything...
The nomenclature "2.0" has become synonymous with second generation trajectories. We have all experienced the benefit of the "Web. 2.0" movement, a term coined in 1999 by Darcy DiNucci and subsequently popularized at conferences from 2004 onwards. On the surface Web 2.0 suggested a new version of the internet however it was not intended to refer to an update to any single technical specification, but rather to cumulative changes in the way web pages are made and used with particular reference to social media.1
A closer analysis of the 2.0 concept suggests that five dynamics align to create the conditions which trigger a step past a "1.0" trajectory; these include;
1. A better understanding of the limitations and potential of current technology by those closest to it;
2. An adequate flow of investment towards continuous experimentation
3. A (formal or informal) systems integration of previously disparate stakeholders around that new trajectory;
4. Greater transparency of the significant (versus moderate) returns available
5. Advent of individual or corporate leadership accelerating the change.
Let's consider these five forces at work in a familiar example. On my first birthday in 1961 2 the then US president, John F Kennedy announced before a special joint session of Congress the dramatic and ambitious goal of sending an American safely to the Moon before the end of the decade.
"Let us face it, PM1.0's definition has really had an inauspicious start..."
All five 2.0 forces can be seen at work in achieving this clear technological leap. Firstly President Kennedy did not set this new vision without NASA informing him of the limitations and possibilities of 1950's rocket propulsion systems as witnessed in the Gemini and Mercury programs. Secondly and thirdly NASA acted as funder and systems integrator for this goal, managing the most complex of supply chains from life support systems to lunar landing modules and in doing so triggering one of the most rapid periods of continuous experimentation in history. In terms of incentive, the political returns to winning the "cold war" against the USSR were deemed enormous particularly after the shock that a Soviet, not an American, was the first man in space in 1957. Lastly despite the many social and racial problems of the era and potential application of federal dollars, JFK's leadership in setting the new space trajectory set the world alight resulting in Armstrong and Aldrin landing on the moon on July 20, 1969. By setting the Moon landing goal, JFK ensured the world stepped past "space exploration 1.0."
How do these concepts help us assess where we are with personalized medicine?
PM 1.0
First of all we need to contextualize Personalized Medicine or PM 1.0. Let us face it, PM1.0's definition has really had an inauspicious start. Simultaneously slighted by investors and industry leaders as relating to a niche (read minor fringe activity), only for those experimenting in oncology, or confused as an ersatz buzz phrase for everything from sports monitoring devices to regenerative ageing surgery3, in the first part of the last decade PM was often over hyped and constantly reshaped in favour of someone else's preferred definition. I have personally witnessed the targeted therapy and companion diagnostic space been subjected to constant (and frankly confusing) renaming with terms like theranostics, pharmacogenetics, personalized healthcare, precision medicine and stratified medicine. More telling perhaps has been the label (communicated up to the "C" suite and from there to investors) from many close to the pharmaceutical science that "the science is not there yet". A phrase used to correctly suggest that we still have a long way to go to understand one (and only one) dimension of PM namely the genetic and molecular underpinnings of personalizing treatment.
Our own in-depth analysis of PM 1.0 suggests that despite recent industrial thawing towards a PM enabled business model, itself triggered by accelerated approvals of biomarker aided therapies4, PM is really still a series of disconnected building sites and stakeholders. Moreover PM 1.0 is often too narrowly confined from within the pharmaceutical industry, to investments in responder testing for targeted therapy. Nor have the incentives for PM 1.0 been clear. Given the financial uncertainties implied by sub setting small patient cohorts for therapies subject to $1bn development fees, the response by many in the Pharma industry is to default to a PM approach versus a one size fits all therapy launch only when the regulatory pathway dictates such a PM approach. Whilst leading companies have supported some degree of central strategic planning and training, our observation is that in the majority of cases where a therapy will be commercialized with a test, it is the FDA or EMEA who are arbitrating the choice of PM on a particular asset versus a CEO led strategic missive.
If we look instead at Payers, likely the only other major industrial player who has the wherewithal to organize an acceleration of PM, the picture is equally reactive. Yes there are payer led initiatives, United Healthcare5 for example currently support up to $.5 bn on genetic testing for their patients. Aetna have had a central PM person in place assessing the space for as long as our company has existed. However by the nature of their industrial architecture payers are highly data driven6 and too often this has been lacking in PM1.0 and has consequently not triggered a payer led drive towards accelerating PM.
"...the forces for PM2.0 and a more radical acceleration of PM are aligning in anticipation of individual or corporate leadership."
In my view, whilst Pharma and Payers will increasingly be willing participants in PM 1.0 this incrementalism will not translate into the radical healthcare change implied (and feasible) by an era of personalized medicine. However our research also suggests that the forces for PM2.0 and a more radical acceleration of PM are aligning in anticipation of individual or corporate leadership. We outline in broad terms our case for PM2.0 below.
The case for PM 2.0.
To assess the case for PM 2.0 with a structured lens we applied the five forces (cited above) aligning for a 2.0 trajectory to personalized medicine and further discuss below.
1. Understanding the limitations and potential of PM 1.0 technology: With some 70 targeted therapies and companion diagnostics ( biomarkers) on the market7 and the almost daily now, translation of genetic research into bedside innovation, the last 10 years have seen a period of rapid learning about the power (and limitations) of the early application of the technologies underpinning personalized medicine. In translational medicine labs across the globe, new insights are being harvested giving us a smarter future perspective of where the "technology can go" even if they are not there yet.
2. Investment in Personalized Medicine experimentation: As recent investment analyses illustrate8 PM is benefitting from an ever higher profile with professional investors as well as industry leaders. This in turn is triggering higher levels of clinical and industrial experimentation incorporating everything from super computers9 to next generation sequencing10. Nor is such experimentation limited to technology , involvement for example of social media and smart phone applications in patients personalized healthcare empowerment is a significant force for change and is symbolic of the disparate stakeholders aligning around the PM concept.
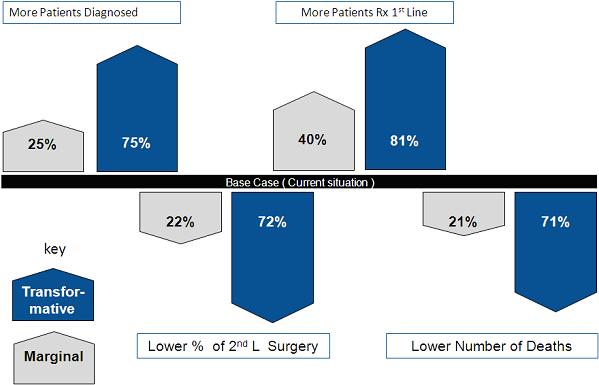
3. Systems Integration in Personalized medicine: Perhaps a little more opaque, nonetheless recent publications have started to argue that PM lends itself to a systems integration approach, as Ginsberg et al argued in a recent JAMA paper " ... to minimize health care cost increases, genomic approaches must replace existing inefficient technologies and reduce the use of downstream resource"11. Our own research, into the transformative capabilities of personalized medicine, suggests a significant clinical dividend is available but only when PM is introduced as part of a total system in a disease. Specifically to replicate a PM 2.0 world we have modelled the combined impact of a systems integration in Melanoma derived from better early diagnostics, prognostic testing and responder testing alongside new first line targeted therapies aimed at reducing second line surgery and the burden of 5 year surveillance, both of which conspire to drive up the cost of melanoma12. Figure one illustrates marginal and transformative clinical impact of a systems integration model applied in one disease area (melanoma). We have replicated these models in infectious and metabolic disease and see similar profound impact.
Figure 1: Melanoma PM 2.0 Clinical Impact Analysis
"...a systems integration approach in PM could also provide significantly more revenue for Pharma and significant cost reduction for Payers than their current business models."
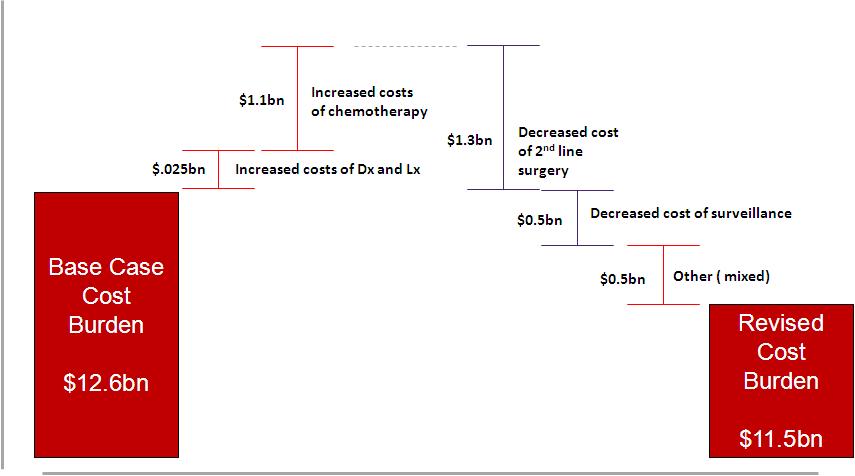
4. Greater transparency of the significant (versus moderate) returns available from PM2.0: Within Pharma there are already the whispers (to be fair Roche have been promoting the investor benefits of PM for some years now) that PM might make economic good sense. In a reversal of the widely held perspective that treating small patient groups meant financial suicide for large Pharma, the triple benefits of accelerated regulatory approval and smaller clinical trials and significant clinical impact for patient subgroups, have already delivered early revenue and time to peak sales opportunities versus the one size fits all model. Using our intervention-based business models we have tried to elucidate better the potential future impact on Pharma and Payer profits of a shift to a PM 2.0. paradigm. What these models point to is that a systems integration approach in PM could also provide significantly more revenue for Pharma and significant cost reduction for Payers than their current business models. Figure two describes the increases and decreases in costs in our Melanoma 2.0 model. We note that both industrial groups (Pharma and Payers) obtain a significant economic dividend from a PM 2.0 aligned program.
Figure 2: Melanoma PM 2.0 Economic Impact Analysis: Melanoma transformational Model shown only
5. Advent of individual or corporate leadership accelerating PM 2.0 change: One of the major gaps in my view mitigating against a PM 2.0 drive to date has been lack of "C" suite leadership from Pharma and Payers. I acknowledge that many CEO's and R&D heads now list PM as a pillar of growth for their business but that is not the same as organizing a business model shift towards PM. Nor do I subscribe to the academic view from the likes of Christenson that PM will ultimately disrupt (threaten) the Pharma and Payer business models in the same way that the PC changed forever the main stream computer business in the 1980's.13 One of the strengths of the Pharma model has been to manage and spread risk in a long cycle business. Take GSK as an example. Andrew Witty has met the challenge of declining R&D productivity not only with new internal innovation process but by promising shareholders that China (and developing countries) is an area for significant growth. Given that only 3% of GSK's revenues in 2012 emanated from China there are decades of opportunity to offset the risks of long cycle therapeutic discovery. GSK's model is well configured to survive without a compulsion to lead personalized medicine into a new trajectory. At the other end of the spectrum Severin Schwan's embrace of PM as integral to Roche's future is either prescient or an outlier to his peers in the industry. The arrival on the disease management scene of a new entrant called Calico, backed by Google and led by a PM veteran in Art Levinson, has the potential to increase the competitive pressure on large industry incumbents but will not competitively impact in the short term without the bold purchase of say Astra Zeneca or one of the other recently troubled Pharmaco's. The bottom line however is that, at the minute, PM does not seem to have its Bill Gates or JFK.
"At the other end of the spectrum Severin Schwan's embrace of PM as integral to Roche's future is either prescient or an outlier to his peers in the industry."
Discussion and Summary
I have tried in this brief piece to apply a slightly different lens to the directions for PM. It is neither the only lens nor the optimum one but it does highlight several truths.
Well organised, PM can deliver the called for healthcare step change and provide the industrial rewards commensurate with (or greater than) other successful investment in healthcare reforms. However despite our perspective that the five forces for a PM2.0 trajectory are aligning, they are not yet in alignment. We are left wondering if the goal for example to consign diabetes to the clinical and economic equivalent to the common cold within the next decade years is impossible, or simply the absence of a singular vision or perhaps visionary.
References
1. http://en.wikipedia.org/wiki/Web_2.0
2. http://history.nasa.gov/moondec.html
3. http://everythingaboutstemcells.com/tag/regenerative-and-personalized-medicine/
4. http://www.diaceutics.com/future-healthcare-personalized-medicine
5. https://home.modernhealthcare.com
6. http://www.futuremedicine.com/doi/abs/10.2217/pme.11.77?journalCode=pme
7. Personalized Medicine Coalition
9. http://www.prweb.com/releases/2013/4/prweb10644264.htm
10. http://www.foundationmedicine.com/about-investors.php
11. Realizing the Opportunities of Genomics in Health Care. Ginsburg GS, JAMA, April 10, 2013—Vol 309, No. 14, 1463-1464
12. http://www.youtube.com/watch?v=D45o_eDu4cc
13. http://www.claytonchristensen.com/books/the-innovators-dilemma/ and http://www.claytonchristensen.com/books/the-innovators-prescription/
About the author:
As Chief Executive Officer of Diaceutics, Peter has led the transformation of Diaceutics from a personalized medicine consulting firm to a highly focused business consulting, laboratory and educational services and software application firm specializing in personalized medicine. Peter recognized early on that personalized medicine would be an increasingly large part of the drug development paradigm, representing a significant shift in how the pharmaceutical industry operates. In response, Peter combined the need for a standardized and structured process to effectively commercialize and launch companion diagnostics alongside drug development with a method for institutionalizing personalized medicine by envisioning and creating a toolbox of personalized medicine specific software applications and new fit for purpose services.
In his role at Diaceutics, Peter brings over twenty-four years of experience in international healthcare, having directly initiated and managed the launch of four pharmaceutical, seven OTC and five diagnostic products in addition to three joint ventures and two major joint corporate marketing campaigns. In addition to Peter's extensive operational experience in the US, he also has substantial experience running operations in all the key healthcare markets of the world, including Germany, the UK and Japan.
Peter has spent two extended periods in applied industrial research, including a year at MIT's Pharmaceutical Program at the Sloan School of Management in Boston.
Does personalised medicine make economic good sense?