News
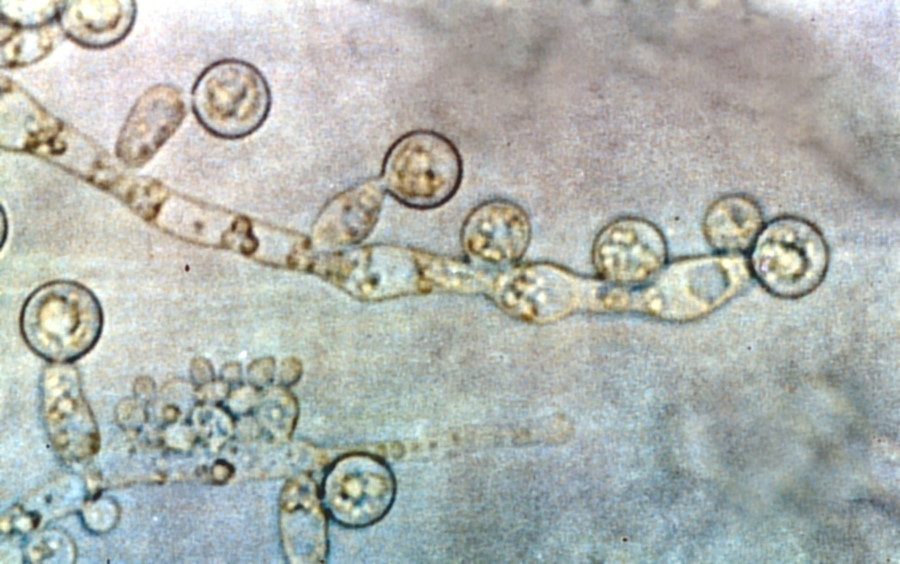
FDA advisors back Cidara antifungal rezafungin
An FDA advisory committee has recommended approval of Cidara Therapeutics' once-weekly antifungal therapy rezafungin, setting it up to become the first new option for severe infections caus




