Ping An completes trial of AI-based retinal disease screening system
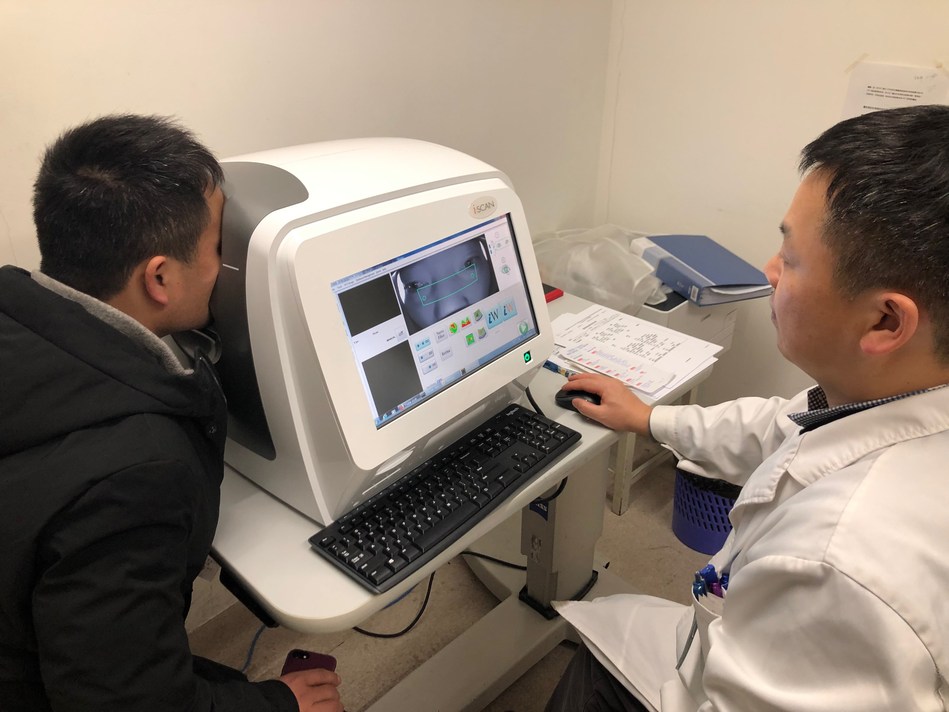
Ping An Technology has announced it has completed a clinical trial of the world’s first intelligent optical coherence tomography retinal disease screening system.
The technology arm of China’s Ping An Insurance Group is developing the system jointly with US-based Optovue.
It is the first artificial intelligence (AI) imaging and screening system in the world that integrates an OCT retinal imaging device and AI lesion detection software.
The clinical trial was conducted at three well-known research institutions, the Eye and ENT Hospital of Fudan University in Shanghai, Shanghai First People's Hospital of Shanghai Jiao Tong University, and Shanghai Tenth People's Hospital of Tongji University.
OCT is a high-resolution, non-contact and non-invasive diagnostic technique that renders an in vivo cross-sectional view of the retina.
The intelligent OCT retinal disease screening system developed by Ping An Technology focuses on the integration of an OCT retinal examination device with AI-powered screening software and applies the Generative Adversarial Network (GAN) technique in the model training process.
The clinical trial was initiated in December 2018 at the three research institutions and involved 784 study subjects.
From the start of an OCT examination to a patient scan code used to generate an intelligent screening report, the entire process can be completed in three minutes.
The findings suggested that the accuracy of tests on the samples by the system in image quality assessment, lesion detection, and referral urgency assessment were 99.2%, 98.6%, and 96.7% respectively, comparing favourably to baseline data provided by experts.
Based on the findings the company said that incorporating AI into screening is important to detect eye diseases earlier and allow them to be treated to allow for maximum benefit from any therapy.
The company said that the results so far suggest that “AI will be extremely helpful to medical institutions in the future”.