Biotts claims a first with insulin delivery across skin
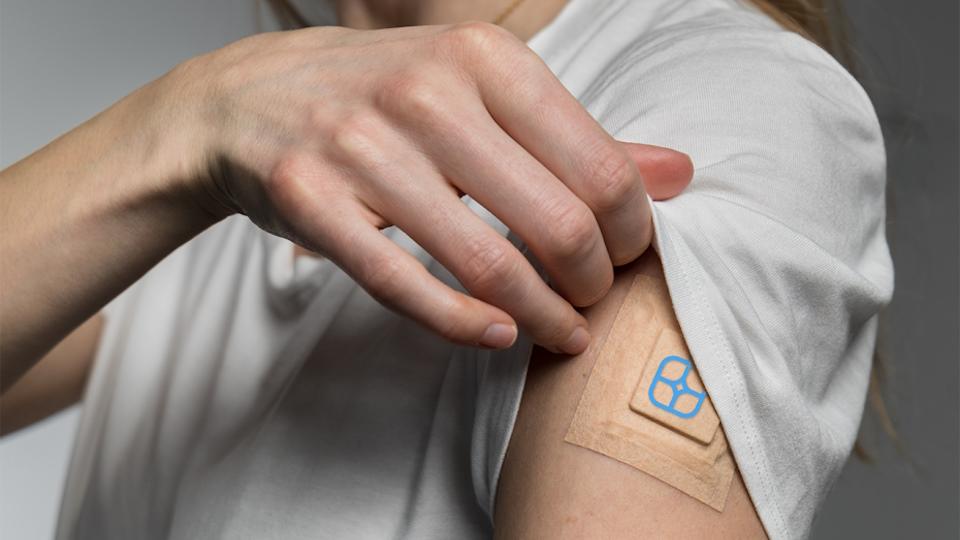
Polish drug delivery company Biotts says it has demonstrated delivery of insulin across the skin for the first time without the use of an injection system.
Transdermal delivery of insulin is not new, but so far has focused mainly on the use of microneedles, sometimes made from biodegradable materials, to overcome the skin’s natural resilience as a barrier to large molecules. Various microneedling technologies have been approved by the FDA, but none have so far been cleared for use with insulin.
Biott’s non-invasive MTC-Y is a mixture of excipients that form a semi-solid carrier that transports insulin across the skin into the blood and – according to animal studies – delivers longer-lasting and more stable levels than subcutaneous injections, without breaking the skin layers.
The prototype transdermal patches used in the animal studies were able to deliver insulin at therapeutic doses for up to five days, which could save up to 20 injections for someone with type 1 diabetes. Moreover, the patch did not need to be worn throughout that time, delivering its payload within a few hours.
Last year, Biotts partnered with insulin producer and diabetes care company Bioton, also based in Poland, to develop the transdermal insulin product for human use, and say they are ready to move onto the next phase in the hope of bringing it to market from 2027 onwards.
Biott’s chief executive, Jan Henk Hendriks, said that the proof-of-concept study offers the potential for a transdermal insulin product that can deliver “better and more comfortable treatment, without an increase in the cost of therapy, and without generating excess medical waste.”
He noted that the result is especially meaningful considering the impact injection burden can have on children with type 1 diabetes, and the rising incidence of type 2 diabetes worldwide. According to a report by the International Diabetes Federation (IDF), the number of people aged 20-79 with diabetes will reach 643 million by 2030.
“Not only will this improve the quality of life of millions of diabetes patients around the world, it will also inspire the pharmaceutical industry to develop similar alternatives in other therapeutic areas with our technology,” added Hendriks.
Biotts has already run a phase 1 trial of a transdermal formulation of dapagliflozin, the active ingredient in AstraZeneca’s blockbuster diabetes, heart failure, and chronic kidney disease (CKD) therapy Farxiga/Forxiga. The patch product would be worn over seven days, doing away with the need to take a daily oral dose of the drug.
Animal studies have also shown the feasibility of using an ointment formulation of semaglutide, sold as Ozempic for type 2 diabetes and Wegovy for weight loss by Novo Nordisk, as an alternative to a weekly injection.
Novo Nordisk also spent several years trying to develop an oral formulation of insulin that could replace injections, but abandoned the project in 2022. Meanwhile, another oral insulin tested by Oramed Pharma was dropped last year after a failed phase 3 trial.