mHealth Monthly Mashup: Release 24.0 – “Let’s Get Our Mobile On!” A Pharma Call-to-Arms

As our health technology focus month draws to a close, this month’s mashup looks back at an incredible year for mobile health, and forward to a bright pharma future…
What a year for mobile health! We had plenty of fits and startups, high tech and low dreck, commercialization, innovation, and finally regulation. Many steps forward, a few back, the industry skittering along while trying to get its bearings, everyone seemingly clear about where we ultimately need to go, but nobody quite sure how to get there. Tap, tap, tap...
So let's take a look at some of the significant milestones of 2013, and discover signposts for an amazing year ahead. Pharma is poised to take full advantage and certainly understands the many benefits, but for many good reasons remains cautious. Millions of physicians, patients, and their caregivers are already there regardless, so let's get our mobile on!
Lucky 2013: A Transformative Year for mHealth
Given the exponential rate of mobile expansion, Moore's Law must apply with gusto, not only the technological capabilities doubling every two years, but the behavioral, institutional, entrepreneurial, and governmental influences as well. Across every category the rallying cry is clear: Respect your audiences, heed the limitations, yet embrace the inevitable and dive in.
"New regulation, exciting app accomplishments, and tantalizing mhealth trends drove the year of frenetic growth and change in the space."
New regulation, exciting app accomplishments, and tantalizing mhealth trends drove the year of frenetic growth and change in the space. Together they form a composite of a digital health future tirelessly buffeted by such surprises as United Healthcare buying Audax Health and Google selling Motorola. Never a dull moment indeed! Are we having fun yet?
Mobile Milestone 1: Draft Guidance Becomes "Final"
Who knew that the FDA would keep its promise to finalize draft guidance for mobile medical apps by the end of fiscal year 2013, and actually issue them in September? The long-awaited, two-years-in-the-making "nonbinding recommendations" add welcome clarity and a framework to an otherwise confusing and chaotic space, ostensibly setting boundaries.
Bradley Merrill Thompson—a legal expert on FDA regulatory, reimbursement, and clinical trial issues whom we interviewed when the draft guidance was first issued—defines in his eminently readable and thorough mobihealthnews "FDA Regulation of Mobile Health" the essential criteria for whether a mobile health app is subject to regulation:
1) If the app acts as an accessory to an already regulated medical device, or
2) If the app turns a mobile platform (such as a tablet or smartphone) into what is already defined as a medical device
Mr. Thompson and most experts seem generally pleased with the delineation and FDA approach, as the guidance not only provides direction but expresses the agency's willingness to work transparently with the industry to answer ongoing questions and ever-changing needs. A website and email address are even available to solicit and analyze input.
By clearly defining what the FDA designates as a "medical device," the agency balances public safety with ongoing innovation by drawing the line at level of risk to patients: High to low, from an app that has or facilitates the ability to diagnose and even treat a disease, to one that merely tracks and manages lifestyle, health and wellness, and other non-clinical tasks.
The good news is that distinction makes a vast majority of apps immune to regulatory oversight. Of the hundred thousand or so available apps, only 103 have been approved by the FDA so far, with a limited number in the queue. But the bad news is that focus on "intent of use" results in lack of clarity regarding the definition of "disease," "device," and "accessory."
"So what is pharma to make of all this? And how does a brand or franchise team move forward in the space?"
So what is pharma to make of all this? And how does a brand or franchise team move forward in the space? Clearly the implications are complex, but the mood is optimistic—and meanwhile our healthcare audiences have already officially gone digital and mobile, and have expectations of their own. But precedent has already been set. Let's take a look...
Mobile Milestone #2: Pharma Innovation & Experimentation
Despite deep-rooted and ongoing regulatory and reputation management concerns, seismic technological shifts, and shrinking budgets, pharma has resolutely and often boldly explored the space throughout 2013. Across every category from corporate to patient ed to branded, pharma launched great stuff throughout the year, highlights featured on FierceBiotechIT.
Some favorites for the year include: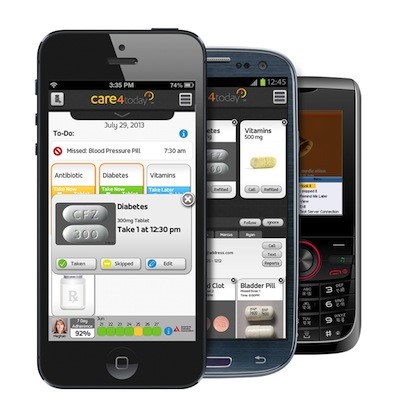 Janssen Healthcare's Care4Today mobilizes patient adherence across multiple disease states with innovative and highly interactive approaches to positive reinforcement through charity and family support. The app ties into broader compliance and health and wellness programs including exercise, education, and live support.
Janssen Healthcare's Care4Today mobilizes patient adherence across multiple disease states with innovative and highly interactive approaches to positive reinforcement through charity and family support. The app ties into broader compliance and health and wellness programs including exercise, education, and live support.
 Genentech's "Killer Muenster" tantalizes kids with an irreverent but informative game replete with visual puzzles that educate about DNA, the double-helix, genetics, and health. Interesting websites, a YouTube channel, online swag store, and other touch points create a surround sound multichannel digital experience that tells a compelling mystery, all accessible from a cool iPhone app.
Genentech's "Killer Muenster" tantalizes kids with an irreverent but informative game replete with visual puzzles that educate about DNA, the double-helix, genetics, and health. Interesting websites, a YouTube channel, online swag store, and other touch points create a surround sound multichannel digital experience that tells a compelling mystery, all accessible from a cool iPhone app.
Revisiting our friends at POCKET MD, we find hundreds of other recent pharma mobile apps from numerous functional categories covering branded content, disease ed, games, clinical trials, calculators, trackers, CME, sales and practice tools, etc. Do some now potentially fall under FDA guidance? Most likely. But the sheer depth and breadth is impressive.
 Consider their dynamic listing of most downloaded apps, and you get a flavor of our pharma mobile health times, companies from Pfizer to Allergan, Novartis to BMS and United Therapeutics creating apps that cover the gamut from dosing to diagnostics to full-fledged consulting services, developed for professionals and patients, branded and unbranded:
Consider their dynamic listing of most downloaded apps, and you get a flavor of our pharma mobile health times, companies from Pfizer to Allergan, Novartis to BMS and United Therapeutics creating apps that cover the gamut from dosing to diagnostics to full-fledged consulting services, developed for professionals and patients, branded and unbranded:
Mobile Milestone #3: Hints of Future Trending & The Quantified Self
If the plethora this past year of conferences, speaker events, and symposia was any indication, mobile health is exponentially expanding in terms of professional, media, and entrepreneurial focus. The startup list curated by Rock Health shows an upsurge in mobile health incubators, while angel investors range across verticals from tech to finance.
So our first inexorable trend in 2013 carrying into the new year is influence and money: Important movers, shakers, and big decision makers from managed care behemoths to hospital groups, universities to consumer goods companies are designing, developing, distributing, and innovating their own mobile health apps, systems, and products.
And while all these players grapple with meaningful use, improving patient and professional engagement, big data, electronic health record integration, chronic disease focus, third world implementation, native vs web-based approaches, and seemingly countless other challenges and opportunities, technological and creative innovation continues undaunted.
 Which leads us to our next big trend to spill into 2014, the road to the Quantified Self, paved by tech that begins to destroy the boundary between the device and the user. Most recently exemplified by Google's fascinating glucose-measuring contact lenses, a smart lens the tech giant hopes to get approved by the FDA and released as an astonishing boon to diabetics.
Which leads us to our next big trend to spill into 2014, the road to the Quantified Self, paved by tech that begins to destroy the boundary between the device and the user. Most recently exemplified by Google's fascinating glucose-measuring contact lenses, a smart lens the tech giant hopes to get approved by the FDA and released as an astonishing boon to diabetics.
But even more intriguing than the sheer technological artistry is the underlying goal: That of eliminating the biggest impediment to the adoption of mobile health and most other user interfaces for that matter—the need for passive rather than active data input. Pop the contacts in, and your glucose level is automatically measured, look ma, no glucometers!
So with that insight comes the third and perhaps most compelling and lasting trend, inchoate at present yet infinite in beckoning potential: The trumping of technology by intent. Coming full circle from the new FDA guidance that bases compliance on what an app does rather than how it's built, the future rests in the human side of mobile health.
"Every stumbling block, inspirational idea, or jaw-dropping possibility in mhealth is dependent on the ghost in the machine..."
Every stumbling block, inspirational idea, or jaw-dropping possibility in mhealth is dependent on the ghost in the machine: Privacy and security hinge more on cultural tolerance than tech; investment decisions are based more on faith than science; while unleashing the power of Big Data is all about who owns it, and what we allow done with it.
Toward a mobile pharma future
Betwixt and between all this stands the elephant in the pharma living room: The decision when and how to play in the mobile health space. Based on these trends the pressure is intense, the benefits immense, and the risks ongoing and ever-changing. But if the industry stays focused on the mission of advancing public health, intent and action will remain clear.
And intent, after all, ultimately supersedes the technology. As devices become increasingly wearable to the point they become part of our bodies, and pentabytes of otherwise lost data are recorded, analyzed, and used to improve health and wellness, pharma devices patients download will become as if not more important than the complex molecules they ingest.
Many of the prediction about mobile health made five years ago are already playing themselves out. My personal prediction is that within a generation or two and at most three, physicians will not only be prescribing more apps than drugs, but like IBM that evolved from mainframes to consultancy, pharma will transmogrify from chemists to data geeks.
What do you see ahead in 2014? And how do you think pharma can best play in the mobile health space? Along the way, let's embrace the mhealth call-to-arms, especially since our constituents demand it, and the clock is ticking for a state change in how we diagnose, treat, manage, and ultimately prevent disease—all with the help of data, not drugs.
About the author:
Michael Spitz is VP of Strategy at Klick Health, where he combines his passion for technology with more than 15 years of clinical content expertise to help engineer digital healthcare solutions. Follow @SpitzStrategy on Twitter for his daily – often hourly – updates on all things digital for the ultimate benefit of patients worldwide.
Closing thought: What do you think is the commercialization of mobile health?











