Every voice matters: demand assessment in rare diseases

With smaller numbers of people affected by a rare disease, traditional market research approaches for assets in this field can be less effective. Duncan Munro describes a FMCG approach that is gaining traction in the pharma industry for these complex decision processes.
The need for pharma to accurately assess demand for a new product in order to understand how it will behave in its target market extends across all disease and treatment areas. This market research objective is typically addressed with large-scale quantitative research, which often includes conjoint-based trade-off methodologies.
However, in the world of rare diseases and early stage assets, itself a significant growth area in the industry, large-scale quantitative approaches are markedly less effective. This is because, despite the fact that rare diseases afflict an estimated 400 million people worldwide, the number with an individual rare disease is often measured only in the thousands, hundreds or even tens of patients. With early stage asset research often targeting a correspondingly small number of highly specialised experts, the opinion of each of them becomes more important.
There are two important features of rare diseases that have a major impact in any choice of research approach. First, any sample collected will typically be insufficiently robust for any technique that relies on aggregated, rather than individual level, data. Second, even a small sample often represents a significant proportion, often greater than 10%, of the prescribing universe for that disease.
SIMALTO
This points away from the more traditional trade-off methodologies of Choice Based Conjoint (CBC) and Discrete Choice Model (DCM) and towards one widely used for over 30 years in FMCG, B2B and service industries, but only recently gaining traction in pharma. SIMALTO, literally meaning Simultaneous Multi-Attribute Level Trade-Off, is a highly flexible methodology for analysing and predicting customer wants, needs and preferences in complex decision situations.
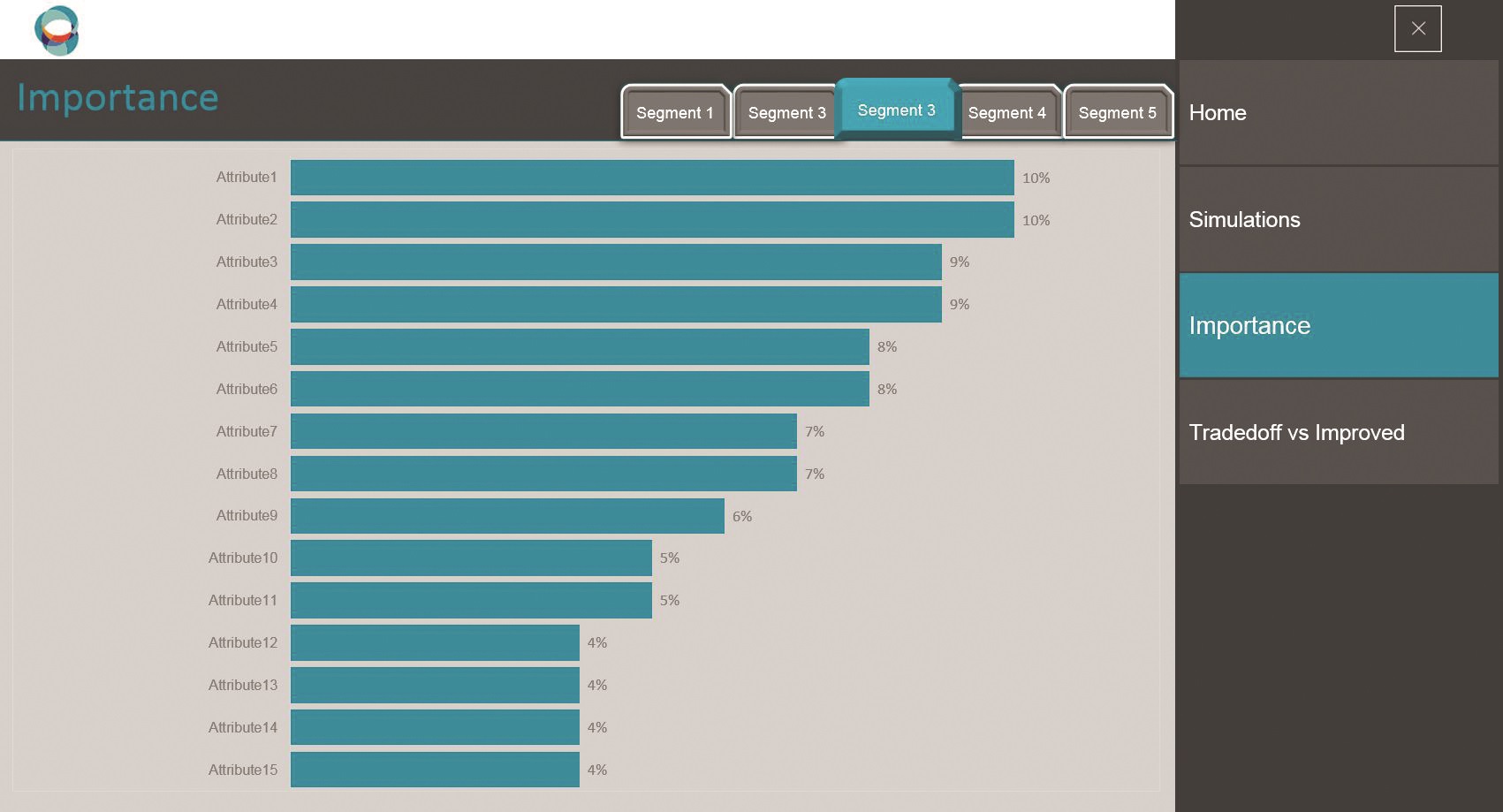
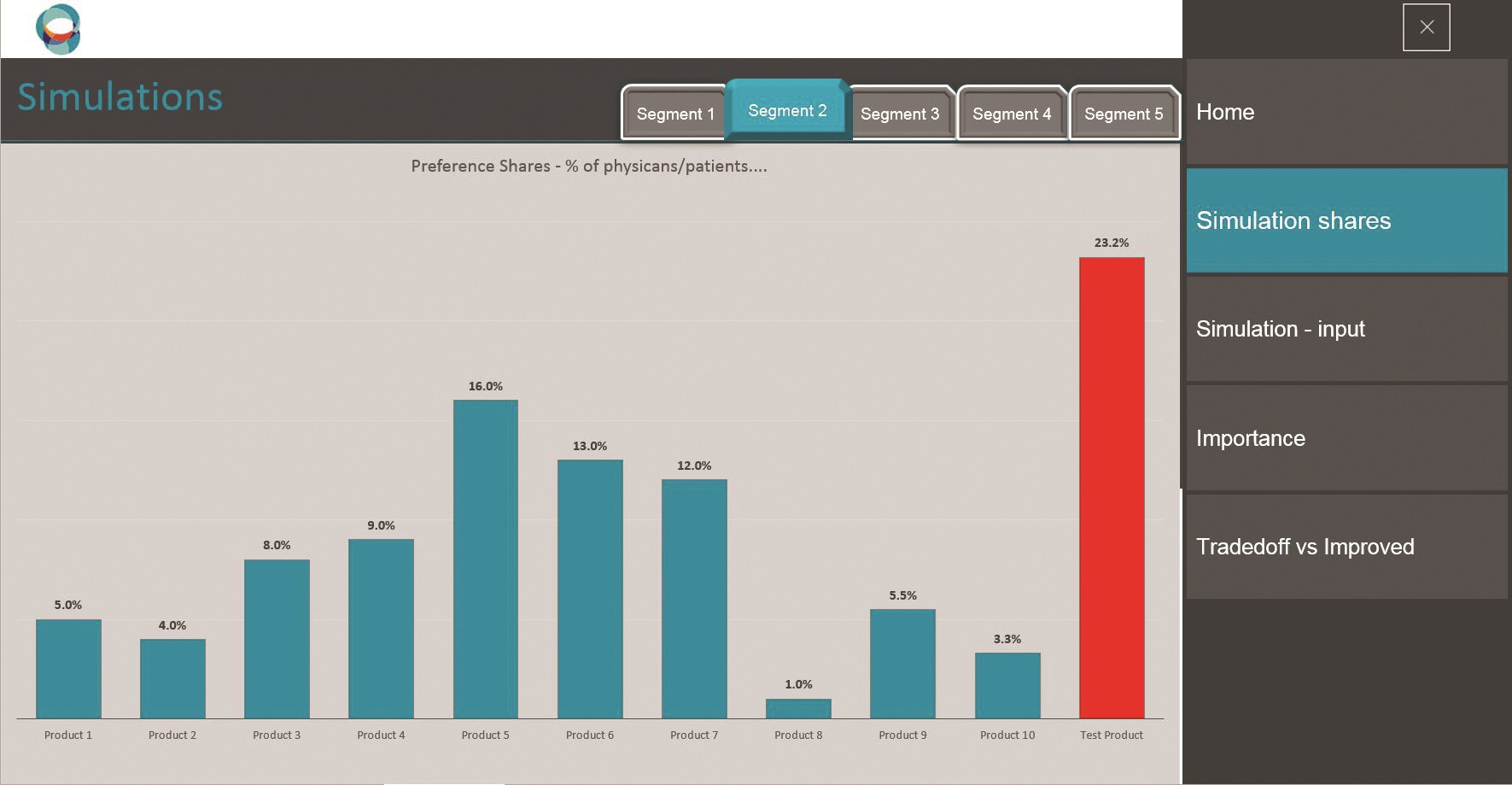
SIMALTO simulator sample screen shots.
A defining feature of SIMALTO is that each respondent reviews and provides feedback on a grid which encompasses a deliberately chosen set of key attributes and their associated levels. Unlike conjoint approaches, which deconstruct the grid, SIMALTO asks respondents to interact with the attributes directly. While it could be argued that this makes the individual task for each respondent more demanding than an equivalent conjoint task, in rare diseases, where respondents hold a very high level of expertise, this can actually be seen as an advantage. This is because each individual is more than a potential expert in a particular condition and best placed to provide input and influence in the ongoing development of any treatment. They are also best placed to factor in the wider disease burden, which extends to family, friends, care groups and broader society and therefore provide a more fully informed perceived and actual value of any treatment.
Moreover, because SIMALTO relies on the respondent’s interaction with the whole attribute grid, it provides preferences and sensitivity thresholds to adopt a new product at an individual respondent level. These can then be interrogated by a SIMALTO simulator to understand the appeal of any alternative product profiles (e.g. upside and downside profiles) that can be described by the attribute grid.
SIMALTO also benefits from relatively light analytical demands when compared to conjoint analysis, which means that the time and effort required to analyse a set of SIMALTO outputs is significantly reduced. This makes it a useful approach when timelines are tight, such as when evaluating a potential early stage asset acquisition. And, in the world of rare diseases, where early engagement can have profound impacts on potential asset development cost savings, as well as improved patient outcomes, in terms of quality of life, each individual voice needs to be heard and understood as clearly as possible.
SIMALTO in use
In one recent example a client wanted to understand the potential opportunity for an existing product in a new and unusual indication in a very different therapeutic category. Interviews were conducted with a small number of specialists in key markets (12 per country, 96 interviews at global level) to understand their acceptance of this new therapy. SIMALTO was used to determine each specialist’s individual threshold levels across six different attributes to construct a simulator, which provided sensitivity analysis, allowing the client to model the potential of a wide range of different alternative profiles for the product. This, in turn, enabled the client to adjust its forecast and select the most appropriate phase 3 trial design to support the brand’s development in this new indication.
Another example involved a client seeking to understand how a new product might perform across a range of different, but related, indications being treated by a range of specialists. Market research was conducted with a small sample of specialists treating each indication (15 per indication). The goal was to understand not only the potential for the new asset in each indication and prioritise its development to address the greatest needs, but also to identify whether the optimal profiles required for each indication would be compatible. SIMALTO research allowed the company to focus development on the highest potential opportunity.
Conclusion
SIMALTO can address the key challenges in demand assessment in rare diseases and early stage assets because it recognises that, in these scenarios, every voice matters. It also provides full information on the preferences and sensitivity thresholds for each individual respondent in situations where standard trade-off approaches are not possible.
About the author:
Duncan Munro is Joint Head of IQ Practice at Cello Health Insight. For more information visit www.cellohealthinsight.com
Read more from Cello Health:
Effective decision making in early asset development and commercialisation











