Long-acting injectables for biologics: Current landscape and future perspective
Biologics are incredibly powerful and targeted. For these reasons, pharmaceutical companies have invested a lot of time and money over the past 20 years developing medications based on these compounds. Notwithstanding their potential, challenges associated with formulation design and administration have prevented their commercialisation, as LAI formulations.1
Between 2015 and 2018, for example, 30% of all drugs approved by the FDA were biologics, and the global peptide market is expected to reach $50 billion in 2027.2,3 In this regard, several long-acting technologies were approved in the past decades for the delivery of peptides, mostly formulated with polylactic acid (PLA) and poly(lactic-co-glycolic) acid (PLGA). However, there are no commercial LAIs for delivery of proteins.
LAIs for biologics lessen the number of administrations required, while also potentially enhancing their pharmacokinetics and, consequently, their safety and effectiveness profiles. The currently existing technologies (microparticles and prefabricated implants) for development of LAIs for proteins can still be improved by creating liquid, stable formulations that are ready to use and simpler to administer, like that of self-administered Depo-Provera (a contraceptive injection that contains the hormone progestin).
The key factor contributing to the limited effectiveness of LAIs for proteins is the complexity and fragility of these molecules, which exhibit poor chemical and physical stability throughout the manufacturing and sterilising processes needed to generate LAIs on a commercial scale. The potential to deliver therapeutic macromolecules at the target site or near to the affected area is one reason why developing in situ forming depot technologies is of broad interest and potentially could be a game changer.
LAIs for the delivery of proteins
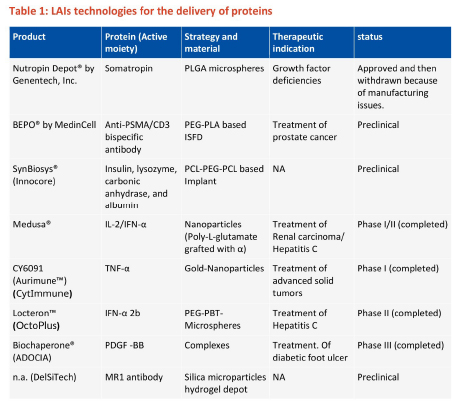
There are no commercial LAI products for delivery of proteins, but many are in development phase. Table 1 summarises some of the clinical trials of different long-acting technologies for delivery of proteins, conducted during the last decade (taken from clinicaltrials.gov). The challenges associated with each of these technologies are described next, followed by a section describing the approaches that can be adopted for successful development of LAIs for biologics.
Challenges with development of LAIs for proteins: Intrinsic properties
The primary reason for the absence of LAIs for biologics (mAb or therapeutic proteins) is their highly complex and sensitive higher order structures and clinical function, which gets distorted during the formulation's manufacturing and storage. Apart from conformational issues, it is mandatory to keep an eye on alterations of post-translational modifications (such as variations in glycosylation patterns, oxidation, and deamidation), which are sensitive to environmental factors like changes in temperature and pH, for example. These problems related to these compounds' inherent characteristics have significantly slowed the advent of their LAIs onto the market.
LAIs for proteins: Challenges associated with manufacturing processes, storage, sterilisation, and lack of appropriate pre-clinical models
Depending on the formulation type and the procedure selected, there could be manufacturing issues in formulating LAIs for proteins. The formulation technique has a more significant influence on proteins than peptides, since they are structurally more complex. The microspheres are made using double emulsion, solvent extraction, or coacervation techniques. The presence of water/organic solvent interfaces in all of these methods results in protein denaturation and aggregation. When it comes to implants, which are generally made by hot melt extrusion (HME), proteins are more adversely impacted by high temperatures and shear pressures than peptides, which lack tertiary or quaternary structure. Forming ISFDs solvents like NMP or DMSO are often used, inducing protein denaturation and loss of their clinical function.
Apart from the formulation strategy adopted, mechanical stress is the key source of protein denaturation when scaling up the formulation. In all the conventional formulation development methods, the molecules and polymers are agitated in reactors during the formulation preparation process, exposing the molecules to shear stresses and air-water interfaces that cause denaturation and aggregation.
Another major source of challenge with LAIs is associated with sterilisation protocols. Filtration-based sterilisation is insufficient for industrial-scale production, since it takes a long time, is not economical, and doesn't always result in 100% sterile goods. As a result, sterilising procedures must be scaled up. However, when scaling up filtration methods, proteins may be subjected to high shear stress when they are channelled through membranes during crossflow filtration, potentially resulting in denaturation. The flow through pumps and valves may also result in micro-cavitation, which creates bubbles that, upon collapsing, create microscopic regions at high local temperatures and pressure that may aid in the production of hydrogen and hydroxyl radicals, as well as the formation of protein aggregates. Due to the sensitive nature of biologics, gamma irradiation and heat sterilisation cannot be employed.
Storage and transportation are also challenging for protein-based LAIs. It is important to keep the protein formulations at the ideal temperature, pH, and lighting during the shelf life. Storage containers also produce contaminations, such as leachables, which might chemically alter proteins, thus losing both their structure and therapeutic function. For the long-term preservation of protein formulations and their eventual delivery to the market, storage temperature is crucial. According to the World Health Organization, 20% of the entire cost of vaccines is accounted for by the cold supply chain, as vaccines and other biologics, such as mAbs, proteins, or even at times peptides, are typically not stable at room temperature and may require storage at 4°C or lower.
Lack of appropriate and robust pre-clinical animal models exhibiting accurate disease pathophysiology is another issue researchers are facing for establishing reliable PK profiles of the formulations. Data generated (safety profiles, toxicity, distribution) through in vivo studies in small animals (rodents) is not sufficient to establish the potency of the formulations, and there is a need to conduct the preclinical studies in large animals (beagle dogs, cynos) for more reliable data.
Future perspectives for developing LAIs for biologics: Solutions for improving delivery systems
The focus right now should be on creating gentle yet robust manufacturing processes and secure, biocompatible polymers (materials) that can be utilised to create nano- or microparticles, implants, and ISFDs.
Batch procedures are used in traditional techniques to produce particulate systems (micro/nano). For large-scale manufacture of the particles, microfluidic techniques should be used, since they use less energy, are reproducible from batch to batch, and preserve the uniformity of the formulations. Moreover, more reliable particle characterisation techniques must be developed to accurately calculate the physicochemical characteristics of the particles. Health Authorities must provide established procedures to evaluate characterisation and toxicity. Synthesis and designing of the polymer platforms which are safe for biologics, enhance their encapsulation/loading, and do not induce acidic pH microenvironment will greatly benefit commercial LAIs, also.
While developing in situ forming depots, future perspectives should be focused on further improving formulation properties, such as reducing viscosity that favours the administration through thinner needles. Also, replacing organic solvents with green solvents will circumvent potential toxicity problems. Researchers should also put emphasis on the development of new bioresorbable polymer scaffolds that allow controlled delivery of biologics.
Outsourcing the development packages and choosing the right partner
Before beginning the project on biologics production and scale-up, the developers should carefully assess their capabilities for small-scale, pilot-scale, and commercial-scale, GMP-compliant production of LAIs. In circumstances where enterprises are unsure of their capabilities, outsourcing the whole project - or the most time-consuming or operationally hard elements of the project - to trusted and well-established CROs with extensive domain experience might be the best alternative.
The CROs must have ability to carry out the projects from inception to regulatory assistance for final reach of the product to market. Additionally, the CROs must have advanced infrastructure for formulation development, where small-scale and large-scale manufacturing of various forms of formulations can be executed through expert scientific personnel. Established vendors, who specialise in large-molecule production and scale-up, have experience working with a wide range of molecules and thus understand the risks involved, as well as possible solutions to overcome those challenges. They also have all the modern technologies and expert manpower in place, and solutions to a wide range of logistical and regulatory challenges.
Furthermore, the vendor should have fully fledged services for conducting pre-clinical studies in-house. Availability of AAALAC accredited, GLP-certified vivariums for both small and large animals help in providing robust and reliable pre-clinical data, increasing the formulations’ speed to market.
Conclusions
For patients, oral biologics administration could seem like a breakthrough. Although there are still numerous problems to be solved, industry is still very interested in this area, as seen by the FDA's clearance of many peptide-based technologies. In contrast to peptides, where several LAI medicinal products have been successfully marketed over the past 20 years, the same cannot be said for proteins. There are still issues with these complex molecules' stability throughout production, storage, and delivery. It is necessary to create new formulation methods that prevent situations where the protein may be exposed to and adsorb at interfaces, causing denaturation, loss of bioactivity, and aggregation formation.
Given that more effective ready-to-use LAI formulations are being researched and submitted, it is essential to identify the challenges associated with specific molecules and develop technologies accordingly. One of the ways to ensure successful delivery of LAIs for biologics is to partner with an established vendor that has capabilities for formulation development and DMPK profiling under a single roof and has experience with a range of biomolecules, including difficult-to-express proteins. Ultimately, taking these informed decisions is critical to the successful production and scale-up of LAIs for biologics, as well as for seamless delivery of the new therapy to clinics.
References
- Gonella, A., Grizot, S., Liu, F., López Noriega, A. and Richard, J., 2022. Long-acting injectable formulation technologies: challenges and opportunities for the delivery of fragile molecules. Expert Opinion on Drug Delivery, 19(8), pp.927-944.
- Anselmo, A.C., Gokarn, Y. and Mitragotri, S., 2019. Non-invasive delivery strategies for biologics. Nature Reviews Drug Discovery, 18(1), pp.19-40.
- D’Aloisio, V., Dognini, P., Hutcheon, G.A. and Coxon, C.R., 2021. PepTherDia: Database and structural composition analysis of approved peptide therapeutics and diagnostics. Drug Discovery Today, 26(6), pp.1409-1419.
About the authors
 Dr Satinder Singh is working as principal scientific manager of DMPK at Aragen Life Sciences and has 18 years of diversified experience across various domains of Indian pharmaceutical and biotech R&Ds. He has worked with Zydus Cadila, Cadila Pharma, Panacea Biotech, and Ipca Labs in different capacities.
Dr Satinder Singh is working as principal scientific manager of DMPK at Aragen Life Sciences and has 18 years of diversified experience across various domains of Indian pharmaceutical and biotech R&Ds. He has worked with Zydus Cadila, Cadila Pharma, Panacea Biotech, and Ipca Labs in different capacities.
 Dr Pratima Srivastava is VP of DMPK at Aragen Life Sciences. She has more than two decades of combined experience in research, teaching, and management. She has worked at Lupin and Central Drug Research Institute previously.
Dr Pratima Srivastava is VP of DMPK at Aragen Life Sciences. She has more than two decades of combined experience in research, teaching, and management. She has worked at Lupin and Central Drug Research Institute previously.











