NICE recommends cannabinoid med for severe epilepsy
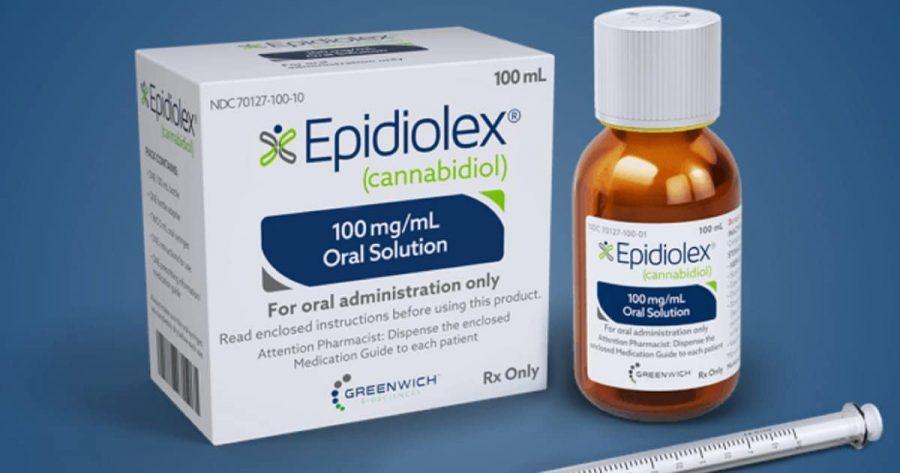
Global biopharma Jazz Pharmaceuticals’ Epidyolex/Epidiolex (cannabidiol), developed by GW Pharmaceuticals and now part of Jazz, has received recommendation (PDF) from the National Institute for Health and Care Excellence (NICE) for reimbursement for adjunctive therapy of seizures associated with tuberous sclerosis complex (TSC), for patients two years of age and above.
Between 3,700 and 11,000 people in the UK live with TSC, a condition that causes mostly benign tumours to grow in vital organs of the body, including the brain, skin, heart, eyes, kidneys, and lungs, and this rare genetic condition is estimated to affect 12 out of every 100,000 people in Europe.
Epilepsy is one of the most common neurological features of TSC and can severely impact patients’ lives. TSC can be diagnosed in infancy, but many aren’t diagnosed until later in childhood, when seizures begin and other symptoms appear.
The NICE recommendation now ensures that all eligible UK patients who may benefit from this medicine will be able to received reimbursed access to Epidiolex from 1st March. Earlier decisions had come from the Scottish Medicines Consortium, Northern Ireland’s Strategic Planning and Performance Group, and the All Wales Medicines Strategy Group.
Epidiolex is a 100 mg/ml oral solution plant-based, prescription cannabinoid-based medicine. It has been approved for use in the UK in three conditions. In July 2019, it was approved by the European Medicines Agency (EMA) and received marketing authorisation under its tradename as an adjunctive therapy for seizures associated with Lennox Gastaut syndrome or Dravet syndrome, in conjunction with clobazam, for patients two years of age and above. Meanwhile, in August 2021, it was approved by the UK Medicines and Healthcare Products Regulatory Agency (MHRA) for use as an adjunctive treatment of seizures associated with TSC in the same patient bracket.
Dr Pooja Takhar, joint chief executive at the Tuberous Sclerosis Association (TSA), said: “The TSA is delighted with this decision, along with people living with TSC and their families. TSC is a very difficult to manage condition, with common issues including epilepsy in eight out of ten people.”
Takhar continued: “Up to half of the people with TSC-related epilepsy are unable to manage their seizures with standard anti-seizure medication, leading to a massive unmet need for new treatment options. This underlines why we are so pleased that this medicine will now be available on the NHS in England, improving lives in the TSC community.”
Simon Newton, general manager at Dublin-based Jazz Pharmaceuticals, added: “This is an important milestone not only for those living with TSC, but also for their families, carers, and clinicians, [demonstrating] the importance of randomised clinical trials and regulatory approval in providing reimbursed access to cannabinoid-based medicines to patients who may benefit.”
GW Pharmaceuticals previously established itself in the cannabinoid science and medicine sector. Founded over two decades ago in response to significant unmet patient need, in 2021 it received the Queens Award for Enterprise in Innovation in recognition of its innovation, leading to regulatory approval of cannabinoid-medicines. In May 2021, the company was acquired by Jazz Pharmaceuticals.
Meanwhile, Jazz’ investigator-led trial of its cannabis extract-based drug Sativex in glioblastoma – an aggressive form of brain cancer – got underway in the UK last year. The three-year phase 2 trial is being carried out by researchers at Leeds University, led by professor of clinical oncology and neuro-oncology Susan Short, to see if adding Sativex (nabiximols) to standard chemotherapy for recurrent glioblastoma can extend survival.











