Experts concerned over mixed results from Takeda's dengue vaccine
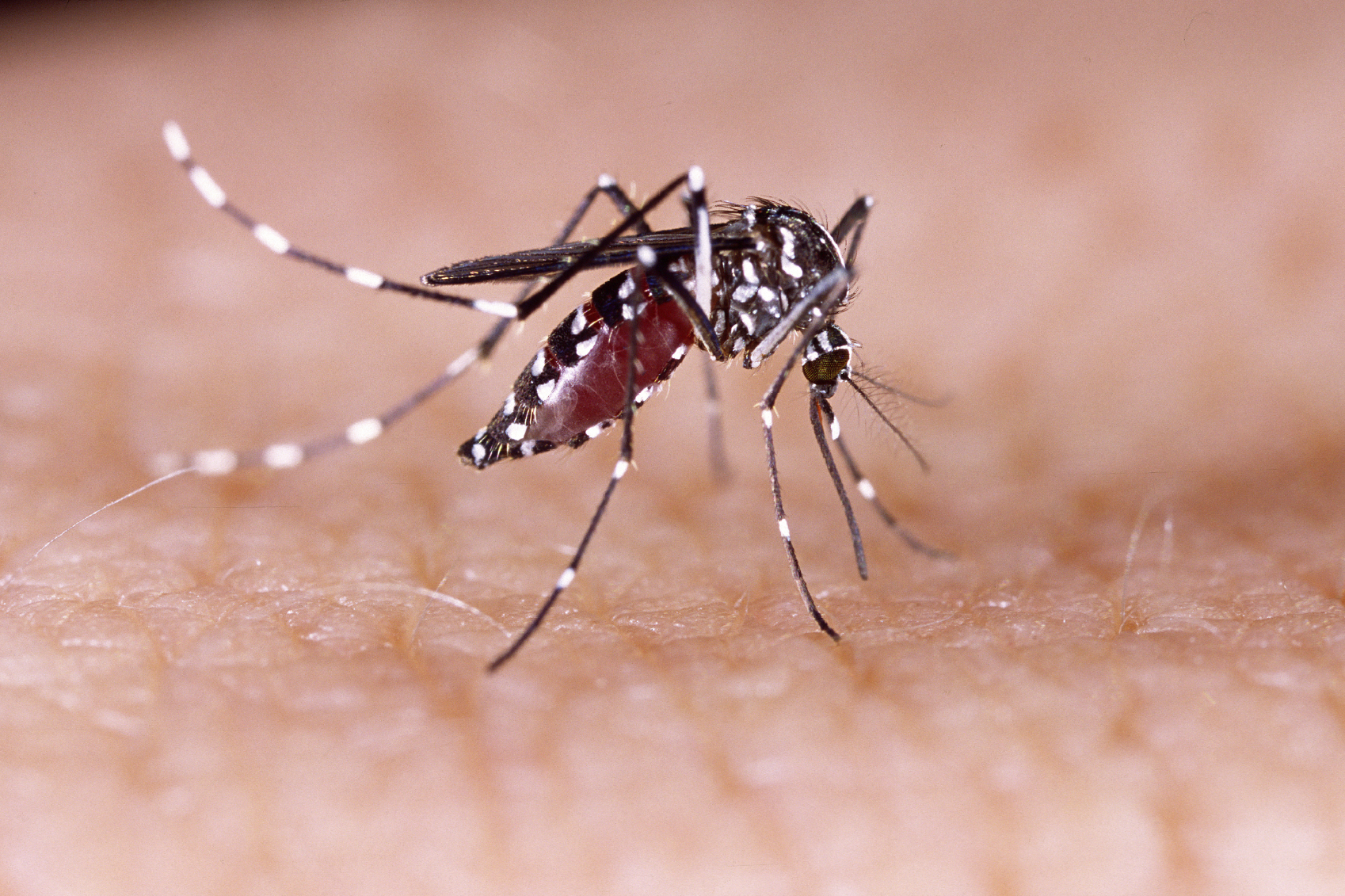
Takeda’s dengue vaccine has met its target of protection against the disease in children – but there are already concerns about mixed results among different strains of the potentially deadly mosquito-borne disease.
There is a pressing need for a new dengue vaccine after a controversy blew up around Sanofi’s Dengvaxia, after the company acknowledged in certain cases it could increase the risk of severe diseases in people who had not been previously exposed.
This limited access to the first ever approved vaccine for the disease after deaths of children were reported in the Philippines, one of the first countries to receive the it.
Officials from Sanofi and the country's health department could face criminal charges over irregularities in the immunisation programme based around Dengvaxia.
Based on results of the phase 3 TIDES study, it looks as if Takeda’s dengue vaccine codenamed TAK-003 is likely to be an improvement on Sanofi’s predecessor.
The trial met its primary endpoint of protection against dengue in children aged four to 16 years, with an overall efficacy of 80.2% in the 12 month period after the second dose, administered three months after the first shot.
Similar efficacy seen in those who were previously infected (82.2%) and those who had not been previously exposed (74.9%).
However Takeda’s statement summarising results pointed to “Serotype-specific efficacy” as all four strains of the disease were represented in the global trial.
Efficacy for serotype 1 was 73.7%, while for serotype 2 the figure was 97.7% - perhaps unsurprising given that the vaccine is based on a live-attenuated serotype 2 virus.
The figure for serotype 3 was 62.6% and there were too few cases of serotype 4 to give an accurate reading, Takeda said.
Dr Anna Durbin of the Johns Hopkins Bloomberg School of Public Health, who has helped develop a rival vaccine with the US National Institutes of Health, told Reuters there could be an “imbalance” in the vaccine, although she noted the overall efficacy was very good.
Dr Stephen Thomas, a dengue expert at the State University of New York Upstate Medical University, who has been a paid adviser for Takeda, Sanofi, and other pharmas, said the variation in type specific efficacy was disappointing considering the need to protect against all dengue virus types.
However he noted that there was no indication of any harm coming from the vaccine.
Takeda plans filings in countries badly affected by dengue beginning in the second half of 2020, and further data are due to be published at the American Society of Tropical Medicine and Hygiene conference later this month.











