As cases rise, WHO says monkeypox risk is "moderate"
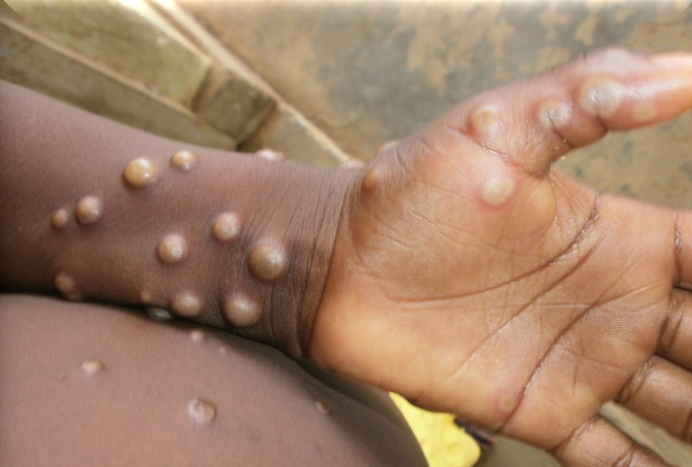
The World Health organisation's latest assessment of the monkeypox outbreak is that it poses a moderate threat to public health, as cases of the infection continue to rise.
In an update posted yesterday, the WHO said it had received reports of 257 confirmed cases of monkeypox from 23 countries, 106 of them from the UK and 49 from Portugal, with another 120 suspected cases being investigated. No deaths have been reported from the infections.
For comparison, there were just seven cases of monkeypox in the UK between 2018 and 2021, according to a just-published paper in The Lancet Infectious Diseases.
The risk level stems from the spread of the virus among people with no established travel links to an endemic area, which is unusual in monkeypox cases, and in "widely disparate…geographical areas," said the WHO.
The big question is whether this change in behaviour is a result of a change in monkeypox virus transmission properties or increased virulence.
"The public health risk could become high if this virus exploits the opportunity to establish itself as a human pathogen and spreads to groups at higher risk of severe disease such as young children and immunosuppressed persons," according to the WHO.
One concern for public health experts is that with vaccination against the similar smallpox virus ending more than 40 years ago, there is little immunity in global populations to help curb the spread of infections.
The priorities for healthcare services should be to provide accurate information to people who may be most at risk of contracting the virus, stopping further spread among those groups, and protecting frontline health workers, said the WHO.
Belgium – with three confirmed cases and three under investigation – has just become the first country to implement a mandatory rather than voluntary 21-day quarantine for anyone who contracts the virus and their close contacts.
At the moment the only approved vaccine for monkeypox is Bavarian Nordic's Jyneos, and the company says it has been inundated with requests for supplies from countries around the world. It says it has the capacity to produce around 30 million doses per year.
Another shot called ACAM2000 from Emergent BioSolutions is licensed for smallpox but could also provide protection against monkeypox if authorised.
Meanwhile, SIGA Technologies' oral drug Tpoxx (tecovirimat) is approved for smallpox, monkeypox and cowpox in Europe, and in the US and Canada for smallpox alone, although it can be used off-label for the other disease. Another drug – Chimerix' Tembexa (brincidofovir) – is approved for smallpox and has shown activity against monkeypox in animal studies.
Monkeypox causes symptoms similar to but milder than smallpox, typically beginning with fever, headache, muscle aches and exhaustion. It is transmitted to people from various wild animals, such as rodents and primates, and is usually a self-limited disease with symptoms lasting from two to four weeks.












