A history of: Mapping breast cancer’s journey from past to present
Breast cancer has been a formidable health challenge across generations, impacting 2.3 million women globally in 2022 alone. However, decades of dedicated research have gradually transformed this disease from an often fatal diagnosis to a highly treatable condition for many patients.
In the US, five-year relative survival rates now exceed 91% - a vast improvement compared to just 73.6% in 1974. Mortality rates have also seen encouraging declines, with a 41% drop in the UK since the early 1970s. In fact, despite accounting for the highest number of new cancer cases worldwide, the World Health Organization no longer ranks the disease as the leading cause of cancer deaths as of 2020.
These milestones represent the cumulative efforts of researchers, clinicians, and patients who persisted through periods of confusion and setbacks. Each new breakthrough unlocked further understanding about breast cancer's biological underpinnings and opened the door to novel therapies targeting its driving mechanisms.
3,000 BCE:
The beginnings of breast cancer research
While the word “cancer” would not appear until much later, breast cancer has been recognised for millennia, with written descriptions and illustrations that match the disease dating back to antiquity. The Edwin Smith Papyrus, an ancient Egyptian medical text from around 3,000-2,500 BCE, contains what may be the first known reference to breast cancer.

Pictured: The Edwin Smith Papyrus. Image credit:: Jeff Dahl - Edited version of Image:EdSmPaPlateVIandVIIPrintsx.jpg,
Unlike it’s other medical papyri counterparts, which solely contained magical spells and recipes, the Edwin Smith Papyrus provides a profoundly practical guide to treating trauma in Ancient Egypt, including 48 case studies detailing traumatic injuries and treatments as observed by Egyptian physicians. Among these were detailed descriptions of several visual characteristics of “bulging” tumours of the breast.
These firsthand accounts reveal how Egyptian physicians over 3,500 years ago grappled with diagnosing and treating this baffling disease using the limited medical knowledge of their era. Their recommended treatment was cauterisation – using a hot instrument to burn away the growths. While brutally ineffective by today's standards, it suggests these ancient doctors recognised that breast tumours required urgent intervention.
460 BCE:
Cancer in an ancient world
Across the Mediterranean – and a few millennia later – Greek physicians, including the “father of medicine” himself, Hippocrates (460 BCE), were grappling to understand the same condition that had befuddled their Egyptian predecessors. Eschewing superstitions as a cause of the disease, believing instead that cancer was a result of natural causes, Hippocrates and his disciples attributed tumours to an excess of black bile in the body, which resulted in localised growths.

Pictured: Vintage crab. Image credit: Free Public Domain Illustrations by rawpixel, licensed under CC BY 2.0.
He hypothesised that, if left untreated, the black bile tumours could spread to other bodily regions through unseen means – an astute early notion of metastasis, despite lacking the biological understanding we have today. Hippocratic physicians went on to characterise different breast tumour types in detail – from hardened, immovable masses fixed to the chest wall to ulcerated, oozing lesions – coining the term "karkinos" (Greek for crab) to describe the tumours' crab-like appearance.
This black bile theory of breast cancer's cause continued to be espoused by the later Greek physician Galen, in 200 BC, who in turn developed the Galenic humoural theory, which claimed that an excess of one of the four “humours” (phlegm, blood, yellow bile, and black bile), would disrupt the body’s natural balance, leading to illness.
16th – 18th Century:
Enter the research renaissance
In Western Europe, the study of breast cancer and medicine in general stagnated during the Middle Ages due to a variety of religious and cultural factors. Instead, many physicians during this period relied heavily on ancient Greek texts like those of Galen, taking them as authoritative and discouraging new inquiry.

Pictured: Al Zahrawi. Image credit: Michel Bakni, CC BY-SA 4.0, via Wikimedia Commons
In contrast, the Islamic world experienced a renaissance in medicine and science during its golden age from the 8th to 13th centuries. Prominent Islamic physicians like Al-Razi (865-925 CE), Avicenna (980-1037 CE), and Al-Zahrawi (936-1013 CE) made important contributions to the detection and treatment of breast tumours. Al-Zahrawi's influential medical encyclopaedia provided one of the earliest known descriptions of mastectomy and cauterisation for breast cancer treatment.
It would not be until the 16th to 18th centuries, however, that alternative theories about breast cancer would gain significant momentum. Following the groundbreaking discovery of the lymphatic system, in the early 17th century renowned philosopher René Descartes put forward a new theory regarding the origins of breast cancer. He proposed that the disease stemmed from issues with lymphatic fluids. This "lymph theory" gained traction when the influential Scottish surgeon, John Hunter, endorsed it nearly two centuries later in the 1700s.
Hunter taught that breast tumours arose from a coagulation or curdling of defective lymph fluid. While this explanation showed little conceptual advancement from Galen's ancient notion of "black bile" causing breast cancer, it may have still encouraged surgeons of that era to more proactively remove lymph nodes in the armpit area that appeared affected during breast cancer operations.
19th century:
The golden age of surgery
Building upon the swath of medical and scientific discoveries that emerged during the renaissance, the surgical field evolved rapidly throughout the 19th century. Thanks to the introduction of radical new systems, including sterilisation, disinfection, and the use of aseptic gloves (elements that are considered baseline standards for operations today), surgery had become a safer practise for patients.
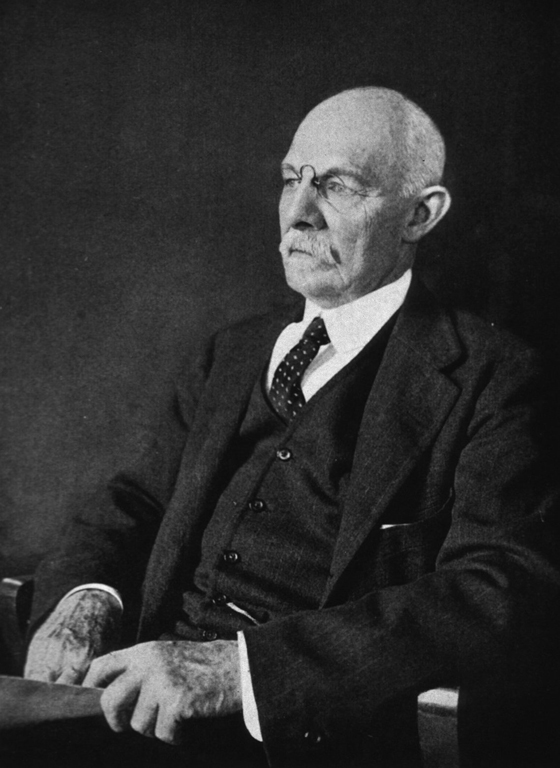
Pictured: William Stewart Halsted. Image Credit: John H. Stocksdale - http://ihm.nlm.nih.gov/images/B14034, Public Domain
Two major breakthroughs during this period include the introduction of antiseptic surgical methods by Joseph Lister in 1867, which reduced post-operative infections, and German pathologist Rudolf Virchow's studies in cell pathology, which laid the groundwork for modern cancer research.
Against this backdrop of medical innovation, pioneering American surgeon William Halsted performed the first successful radical mastectomy (first reported in 1894), an aggressive surgical treatment for breast cancer. While radical, his approach was based on his theory that breast cancer spread primarily through the lymphatic system. By removing the breast, chest wall musculature, and lymph node chains, Halsted believed he could eliminate all sources of local and lymphatic metastasis.
Though disfiguring and intensely invasive, the radical mastectomy offered breast cancer patients a new life-extending option. As such, Halsted's technique became the standard of care for most of the 20th century, despite high rates of recurrence.
Rounding out a century moulded by innovation and discovery, after weeks of diligent research studying a new type of radiation, in 1895 German physicist Wilhelm Conrad Roentgen published his paper "On a new kind of rays". He dubbed these mysterious rays “X”.
1900s-1940s:
Radiation therapy and hormonal treatment
The latter half of the 20th century ushered in major therapeutic advances against breast cancer. Perhaps the most notable innovation to emerge during this period was chemotherapy, which was introduced as a new systemic treatment option. Drugs derived from noxious mustard gas compounds were found to combat breast tumours by disrupting rapidly dividing cancer cells.

Pictured: Charles Brenton Huggins.
Image credit: http://www.nobelprize.org/nobel_
prizes/medicine/laureates/1966/
The turn of the 20th century saw significant advancements in breast cancer research and treatment, despite the prevalence of global conflict and financial uncertainty. In 1913, researchers produced the first chemically-induced breast cancers in animal models, highlighting environmental carcinogens as risk factors.
Throughout the 1920s and 1930s, pioneering surgeons refined and promoted Halsted’s radical mastectomy techniques. A pivotal moment came in 1937 when Charles Huggins at the University of Chicago demonstrated that removing hormonal sources could block breast cancer growth, paving the way for hormone therapy. In the 1940s, radiation therapy was introduced post-mastectomy to target residual cancer cells, and modified radical mastectomies were developed, which were less disfiguring than earlier methods.
By 1949, researchers identified genetic components and inheritance patterns linked to familial breast cancer, leading to deeper exploration of DNA damage, oncogenes, and tumour suppressor genes involved in cancer development.
1950s-1960s:
Chemotherapy and improved surgical techniques
The mid-20th century brought chemotherapy into the arsenal against breast cancer. The impact of sulphur mustards on bone marrow and lymph nodes had been widely observed in soldiers during the Second World War. Noting the destructive potential of the compounds, scientists began to explore the potential of chemical-based therapies.
The initial chemotherapy drugs were derived from nitrogen mustard compounds, which were found to disrupt rapidly dividing cancer cells, including those in breast tumours.
Another breakthrough moment came in 1962, with the introduction of tamoxifen. Originally conceived (no pun intended) as Compound ICI 46,474, tamoxifen was synthesised as part of a project to develop a contraceptive pill. However, instead of supressing ovulation, as was intended, the compound in fact stimulated it. Undeterred, the research team, led by Arthur L Walpole, refocused the failed contraceptive as a way to block oestrogen receptors in some breast cancers. Patients would have to wait to access the treatment, however, as the FDA would not approve AstraZeneca’s Nolvadex (tamoxifen) until 1977.
With the chemical space transforming, it was time for surgery to get a much needed upgrade. In 1971, having spent much of his working life in the cancer research space, American surgeon Bernard Fisher led a landmark cancer trial comparing radical mastectomy with the less invasive lumpectomy – commonly known as breast conserving surgery – combined with radiotherapy. In contrast to the prevailing belief that radical mastectomy was the best course of treatment for breast cancer patients, Fisher found that the two surgical techniques had equal survival rates. Fuelled by his research, Fischer continued to call for better clinical evaluation of treatment options through the use of randomised clinical trials, and lumpectomy officially dethroned Halsted’s radical reign as the standard treatment for breast cancer.
1990s-2000s:
Genetic testing and precision medicine
The 1990s were marked by the discovery of the BRCA1 and BRCA2 genes. For decades, researchers had noted that breast cancer appeared to run in some families, suggesting hereditary factors were involved.

In 1994, after years of painstaking research mapping genetic markers, a team led by scientists at the University of Utah and National Institutes of Health finally isolated the BRCA1 (BReast CAncer 1) gene on chromosome 17. Women with inherited mutations in this gene were found to have up to an 80% lifetime risk of developing breast cancer.
Just one year later in 1995, a second gene called BRCA2 was discovered on chromosome 13 by researchers at the University of Cambridge. Harmful mutations in BRCA2 were also linked to significantly increased breast cancer risk, as well as other cancers like ovarian and prostate.
These two major gene discoveries transformed our understanding of hereditary breast cancers, which account for between 5% to 10% of all breast cancer cases. They paved the way for genetic testing and screenings to identify at-risk women with BRCA mutations.
The isolation of BRCA1/2 provided a genetic explanation for the breast cancer patterns seen in some families over generations. It also opened up new avenues for cancer prevention, such as increased screening or prophylactic mastectomies for mutation carriers.
Elsewhere, the 90s saw researchers build upon the 1974 discovery of epidermal growth factor receptor (EGFR) as a potential breast cancer target. HER2, part of the EGFR family, promotes aggressive tumour growth when overactive, a factor that had been recognised for around a decade before researchers at the biotech company Genentech revealed an innovative treatment approach – a monoclonal antibody that could specifically bind to and inhibit the effects of HER2.
This groundbreaking antibody therapy, dubbed Herceptin (trastuzumab), was shown in clinical trials to dramatically slow the progression of HER2-positive breast cancers where the HER2 oncogene was overexpressed. Given its novel mechanism of action and proven efficacy, Herceptin was granted accelerated approval by the FDA in 1998.
2001-2010:
Shedding light on the once untreatable
The dawn of the new millennium marked a paradigm shift in the way breast cancer was studied, conceptualised, and treated. Building upon the groundbreaking genetic insights of the preceding decade, the 2000s ushered in an era of personalised, molecularly-guided therapies tailored to the distinct subtypes of breast cancer.

In 2000, a landmark study published in the journal Nature used comprehensive gene expression analysis to categorise breast cancers into four intrinsic subtypes – luminal A, luminal B, HER2-enriched, and basal-like. This molecular taxonomy dramatically altered our understanding, revealing breast cancer as a heterogeneous collection of diseases with unique biological behaviours and clinical implications.
Armed with this new lens, researchers could stratify treatment strategies aligned to each subtype's molecular profile and driving oncogenic pathways. Large clinical trials in the mid-2000s validated this approach, demonstrating significant survival benefits when the HER2-targeted antibody trastuzumab was combined with chemotherapy specifically for HER2-positive disease.
As the decade progressed, other novel targeted agents, including lapatinib and bevacizumab were developed to disrupt additional growth signalling pathways in certain breast cancer subtypes. Genomic assays like Oncotype DX also emerged to predict likely chemotherapy responsiveness. However, the particularly aggressive triple-negative subtype lacking hormone receptors and HER2 overexpression remained stubbornly difficult to treat.
A pivotal milestone was reached in 2010 with the completion of the Cancer Genome Atlas' sweeping map of the entire breast cancer genomic landscape across its multiple molecular subtypes. This unprecedented molecular blueprint positioned researchers to continually refine personalised therapeutic algorithms as precision breast oncology charged forward into the next decade.
2010 - 2020:
Genomic profiling continues
Further proliferation of molecularly targeted therapies and genomic profiling strategies to combat breast cancer's heterogeneous biology continued into the 2010s. Several new targeted agents gained approval, expanding treatment options based on tumour subtype.

For HER2-positive breast cancers, additions included pertuzumab, ado-trastuzumab emtansine, and neratinib. The CDK4/6 inhibitors palbociclib, ribociclib, and abemaciclib were approved for HR-positive, HER2-negative disease, blocking cyclin-dependent kinases driving tumour growth.
Immunotherapies like the PD-L1 inhibitor atezolizumab also emerged, though their utility has initially been limited to triple-negative breast cancers with high PD-L1 expression. PARP inhibitors like olaparib and talazoparib reached the clinic for BRCA-mutated breast and ovarian cancers.
On the genomic front, next-generation sequencing and liquid biopsy technologies allowed deeper molecular profiling of breast tumours and their evolution. Multi-gene panel tests like MammaPrint, OncotypeDX, and EndoPredict were adopted to estimate recurrence risk and guide treatment.
Researchers made inroads into breast cancer heterogeneity, identifying novel molecular subtypes like luminal B HER2-enriched. They gained insights into tumour evolution, therapy resistance mechanisms, and microenvironmental interactions fuelling metastatic spread.
However, triple-negative and metastatic breast cancers remained major challenges, driving efforts to develop better predictive biomarkers and combination regimens. Building on this decade's progress, the future promised more personalised, multi-omics-informed breast cancer management.
2020-2024:
Breast cancer research in a pandemic
The COVID-19 pandemic that gripped the world in 2020 had widespread reverberations across the field of breast cancer research, diagnosis, and care delivery.

For patients already diagnosed, the pandemic created further treatment access challenges. Oncologists had to triage cases and adjust treatment regimens where possible to lower infection risks from therapies that caused immunosuppression or required frequent clinic visits. However, the use of neoadjuvant endocrine therapy increased as it allowed some ER+/HER2- breast cancer patients to delay surgery until it could be performed safely.
On the research front, many breast cancer clinical trials faced significant recruitment and enrolment challenges, with patients unable or hesitant to travel to medical centres amid the outbreak. Some trials were forced to pause enrolment or switch to remote data collection and follow-up, slowing the pace of new discoveries.
However, the pandemic also drove innovation in remote patient monitoring and catalysed efforts to improve research access and inclusion for underserved populations disproportionately impacted by both COVID-19 and breast cancer disparities. And, despite disruptions, important new treatments emerged in 2022-2023 with FDA approvals of Trodelvy for metastatic HR+ disease, Tukysa improving survival for HER2+ brain metastases, plus Enhertu and other antibody-drug conjugates, and new targeted therapies like Truqap and Orserdu for metastatic HR+ breast cancers.
While more progress remains to be made, the achievements to date solidify how sustained scientific investigation can revolutionise our ability to detect, treat, and ultimately prevent devastating diseases over time. The journey continues, fuelled by those committed to expanding the boundaries of what's possible.
About the Author
Eloise McLennan is the editor for pharmaphorum’s Deep Dive magazine. She has been a journalist and editor in the healthcare field for more than five years and has worked at several leading publications in the UK.
Supercharge your pharma insights: Sign up to pharmaphorum's newsletter for daily updates, weekly roundups, and in-depth analysis across all industry sectors.
Want to go deeper?
Continue your journey with these related reads from across pharmaphorum
Click on either of the images below for more articles from this edition of Deep Dive: Oncology 2024