How clinical research is zeroing in on fatty liver disease
Americans’ livers are becoming increasingly unhealthy. In the US, non-alcoholic fatty liver disease is now the leading indication for liver transplantation - superseding hepatitis C and, in females, alcoholic liver disease.
Fatty liver disease currently affects approximately 100 million Americans, disproportionately affecting Hispanic Americans. Those rates have increased 133% over three decades, according to a recent study. This rise in fatty liver disease parallels the obesity epidemic, which is driving a variety of metabolic conditions, also including diabetes, lipid disorders, and cardiovascular disease.
Left undetected, fatty liver disease can progress into non-alcoholic steatohepatitis (NASH), which, in turn, can lead to a number of life-threatening conditions, including cirrhosis, liver failure, and liver cancer.
With no FDA-approved therapies currently on the market, care providers have few options to offer patients at the moment. But that may be changing in the near future. Ongoing clinical research has produced encouraging results in several areas, and it seems conceivable that the first FDA-approved treatments for NASH and related liver conditions will come as soon as next year.
The progression of fatty liver disease
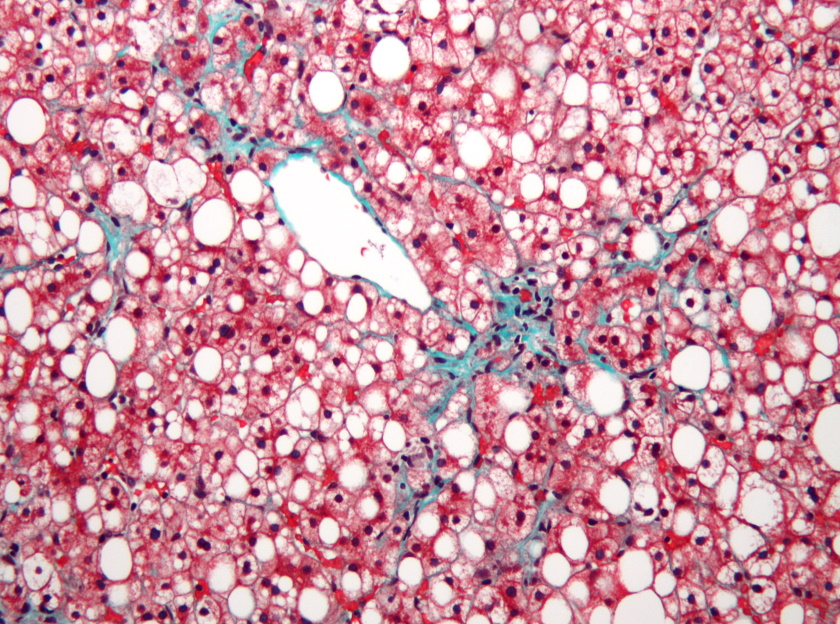
By itself, fatty liver disease is treatable and reversible. However, its symptoms are often silent and non-specific, making it difficult to diagnose until it has progressed into a more serious condition. As fat accumulates in the liver, it creates an inflammatory response - NASH - which in turn causes the liver to try to heal itself through fibrosis, a type of protective scarring.
Fibrosis can be reversed if it’s detected early enough, but as it accumulates, it can lead to the permanent condition of cirrhosis. Cirrhosis can lead to liver deterioration and an increased risk for liver cancer. In these advanced cases, liver transplantation is among the only remaining options.
Progress and challenges in clinical research
However, some promising new therapeutics have the potential to not only reduce fat in the liver, but decrease the inflammatory response that causes NASH and also reduce liver scarring.
Through clinical trials currently in progress, we’re trying to assess these drugs’ effectiveness not just in improving liver health, but also for reducing lipid levels and improving cardiovascular outcomes. There’s a lot of excitement about the potential uses for these drugs, some of which are already FDA-approved for other conditions, such as diabetes and obesity.
The biggest challenge in getting new drugs to market continues to be finding individuals with NASH or fatty liver disease to take part in clinical trials. Patients who look and feel normal can have fatty liver disease, even in an advanced state like cirrhosis, without knowing it.
ObjectiveHealth has one model for addressing this challenge, developing joint-venture, fully integrated research partnerships with GI specialists and primary care providers to offer research at the point of care. Proprietary technology integrates with patient EHRs and provider schedules, using AI algorithms to curate patients with NASH risk factors to identify the highest-risk individuals, and trial-specific matching capabilities help determine the right patients for specific trials using inclusion/exclusion criteria and other data science techniques.
Since blood tests aren't an accurate tool for diagnosis, ultrasound-based technology is relied on for additional testing to characterise the disease more fully, indirectly measuring fat content in the liver, as well as liver stiffness, which correlates with liver scarring. Once a patient’s liver status is more completely characterised, clinical research can be offered as a care solution for patients who opt in. This approach is particularly important for a disease like NASH, where there are no current FDA-approved therapies.
The importance of clinical research
Clinical research offers a patient a higher attention to detail around their condition, important education around the diagnosis and management, and access to new and important therapeutic options that are working their way through clinical trials. One example is Semaglutide, which is FDA approved for diabetes and obesity (and sold under brand names including Ozempic and Wegovy) and is now being studied in fatty liver disease.
ObjectiveHealth recently participated in a clinical trial related to another class of drugs, FGF21-receptor agonists, which target one of the key pathways for fatty liver disease, also.
Where we’re headed
Hepatitis C once affected about 8 million Americans at its peak in the mid-1980s. Research began in the early 1990s and has since reduced that number of cases dramatically, but hepatitis C trials are still going on today to evaluate more effective therapeutics for certain subtypes of disease. So, it can be a very long trajectory for drug development.
NASH, in many ways, is a far more complex disorder, and we're just starting to scratch the surface in understanding how to treat its different subtypes. Although we may soon see the first FDA-approved drugs for NASH, ultimately, there will be many other drugs and drug combinations evaluated as potential therapeutic options for disease variants.
Some investigators believe that NASH will be treated similar to certain cancers, where there are phases of induction, remission, and maintenance. After stopping the active inflammation as rapidly as possible and stabilising the liver, some combination of drugs may be used on an ongoing basis to help maintain the liver in a relatively healthy state and prevent any potential disease progression.
We're entering an exciting new era in terms of metabolic care. As we move away from surgical options like gastric bypass surgery, the emerging therapies mentioned here and others could have a profound benefit for patients - not just for the treatment of fatty liver disease, but also potentially for the entire metabolic syndrome.