US trials of Gilead's remdesivir begin against COVID-19 coronavirus
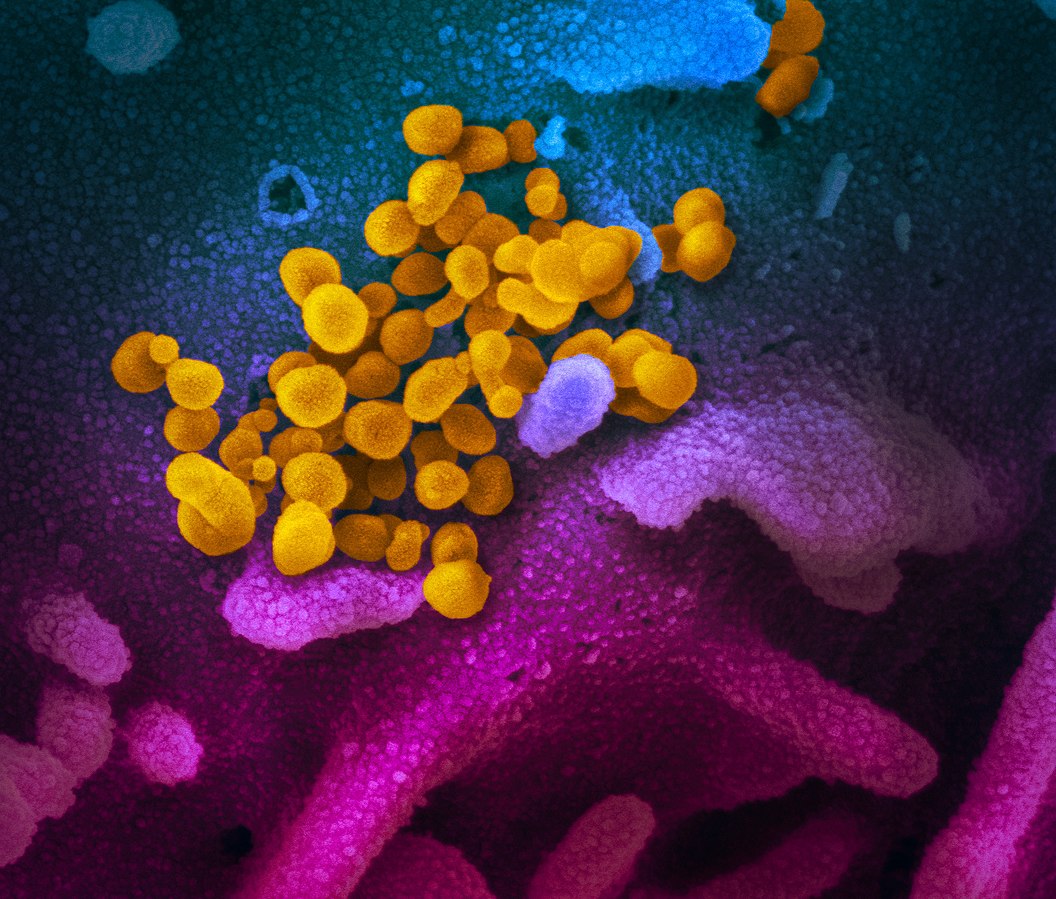
The first US trial of Gilead’s unapproved antiviral remdesivir has officially begun against the novel coronavirus disease COVID-19.
The US National Institutes of Health said the trial will involve hospitalised adults diagnosed with the disease and is taking place at the University of Nebraska Medical Center (UNMC) in Omaha.
It is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH).
The announcement came as the disease began to spread to other parts of Europe from a hotspot in North Italy.
US trials for vaccines against coronavirus are said to be about six weeks away, and are expected to take a year to develop and approve.
In a statement the NIH said the first trial participant is an American who was repatriated after being quarantined on the Diamond Princess cruise ship that docked in Yokohama, Japan and volunteered to participate in the study.
The double-blinded study is led by Andre Kalil, professor of internal medicine at UNMC, and can be adapted to evaluate additional drugs and to enrol participants at other sites in the US and worldwide.
The first setting will be a quarantine unit and UNMC, which also has a biocontainment unit nearby should a higher level of care be needed.
Thirteen passengers from the Diamond Princess were quarantined at the unit on February 17, and since then 11 have been confirmed as having the infection.
Participants in a treatment group will receive 200 milligrams (mg) of remdesivir intravenously on the first day of enrolment to the study.
They will receive another 100 mg each day for the duration of hospitalisation, for up to 10 days total.
A placebo group will receive, at an equal volume, a solution that resembles remdesivir but contains only inactive ingredients.
Clinicians will assign daily scores based on factors including temperature, blood pressure and use of supplemental oxygen.
Participants also will be asked to provide blood samples and nose and throat swabs approximately every two days. Researchers will test these specimens for the virus.
There are no FDA-approved therapies to treat people with COVID-19, the disease caused by the virus now named SARS-CoV-2, formerly known as 2019-nCoV.
Gilead originally developed remdesivir to treat humans with Ebola virus disease, and is a broad-spectrum antiviral that could be used against other diseases.
It has shown promise in animal models for Middle East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome, which are caused by other coronaviruses.
Clinical trials of remdesivir are also ongoing in China. NIAID developed the current study taking those designs into account, and following consultations convened by the WHO on the development of a therapeutic trial for patients with COVID-19.
To be eligible for the NIH-sponsored trial, patients must have laboratory-confirmed SARS-CoV-2 infection and evidence of lung involvement, including rattling sounds when breathing with a need for supplemental oxygen or abnormal chest X-rays, or illness requiring mechanical ventilation.
Infected individuals who have mild, cold-like symptoms or no apparent symptoms will not be included in the study.
China’s Jack Ma Research Foundation has awarded $2.1m towards separate US-based coronavirus therapy research at Columbia University, in New York City.
This will include a project to see whether Gilead’s already approved antiviral sofosbuvir, used in hepatitis C drugs like Sovaldi, could be useful against COVID-19.
Photo: NIAID Rocky Mountain Laboratories (RML), NIH