The Windsor Framework: Insights on the regulatory shift for medicines

The Windsor Framework marks a significant change in the UK's post-Brexit regulatory space, particularly for the pharmaceutical sector. By addressing the complexities of the Northern Ireland Protocol, the framework introduces a UK wide approach to medicine regulation, ensuring that products can move seamlessly across Great Britain and Northern Ireland without dual compliance burdens.
For pharmaceutical companies, this shift simplifies regulatory processes but also demands swift adaptation. The transition to a single UK license, the discontinuation of EU Centralized Procedure authorisations in Northern Ireland, and the mandatory adoption of ‘UK Only’ labelling from January 2025 signals a pivotal change. While these measures aim to reduce supply chain friction, they also require strategic adjustments in labelling, packaging, and pharmacovigilance compliance.
This article explores the key regulatory implications, offering clarity on how businesses can navigate this transformation efficiently.
What is the Windsor framework
The Windsor Framework1 was agreed upon in February 2023 between the UK and the EU to address the key challenges posed by the Northern Ireland Protocol (post-Brexit), ensuring smoother trade between Great Britain and Northern Ireland while preserving Northern Ireland’s alignment with the EU Single Market.
Understanding the Windsor Framework and impact to the regulatory landscape
The key changes2 introduced by the Windsor Framework include modifications to customs and regulatory arrangements, particularly for goods moving between Great Britain (GB), Northern Ireland (NI), and the EU, as well as addressing economic and political concerns in Northern Ireland.
From the regulatory authorisation perspective, all medicinal products currently placed in the NI market are to comply with EU authorisation rules rather than those of the UK (MHRA), and medicines entering NI from GB are viewed as an import from a third country.
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has released updated guidance on UK-wide licensing, pharmacovigilance, labelling and packaging standards, as well as distribution and manufacturing requirements, to ensure compliance with the Windsor Framework.
Impact of implementing the Windsor Framework on human medicines:
- The shift away from EU oversight for the authorisation, approval, and regulation of medicines in Northern Ireland (NI) means that the UK Medicines and Healthcare products Regulatory Agency (MHRA) will assume full responsibility for the NI market. This change enables a single licence and a unified pack bearing a “UK Only” label for medicines across the UK.
- EU Centralized Procedure (CP) Marketing Authorisations will no longer be valid in NI.
- GB only Marketing Authorisations (PLGB) issued by MHRA since the UK left the EU, will be expanded in scope to cover NI.
- The EU Falsified Medicines Directive (FMD) 2011/62/EU requirements will no longer apply to UK medicine packs.
‘UK Only’ labelling and packaging requirements and implementation guidelines
From 1st January 2025, all UK medicinal products must feature a clearly legible ‘UK Only’ label3 on the packaging to ensure consistent labelling and packaging across the UK. This requirement applies to Prescription-Only Medicines, Pharmacy Medicines, and General Sales List products.
When using the ‘UK Only’ label on packaging, the following will apply:
- ‘UK Only’ may be presented anywhere on the outer packaging of the medicine.
- The text must be conspicuous and clearly legible, at least 7-point font and in line with Article 5 of EU Regulation 2023/1182, and other regulatory practice guidance.
- No other font or style requirements are stated.
The table below indicates the eligible territory for the supply of packs before and after 1st January 2025.
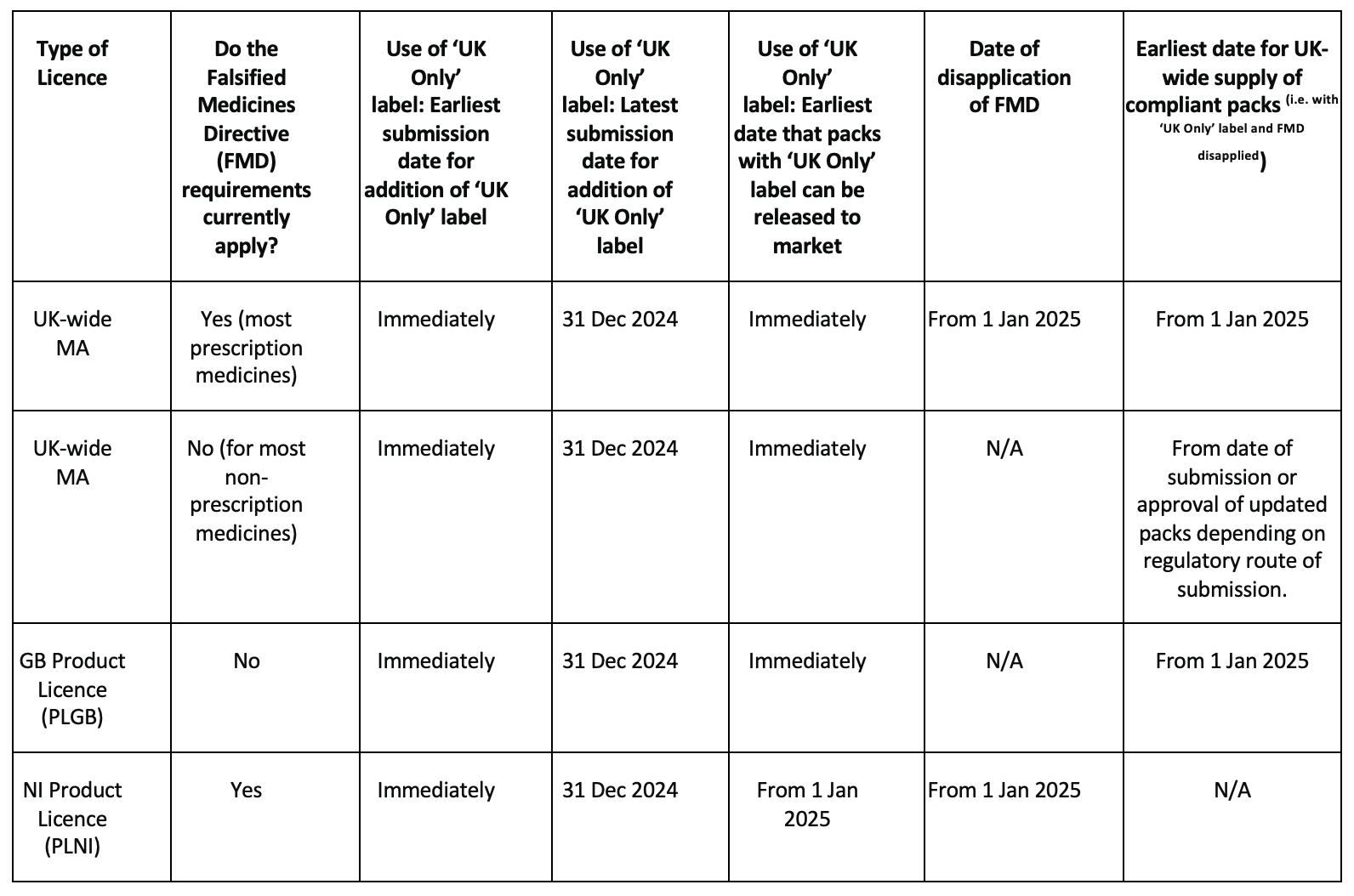
Joint EU/UK packs:
From 1st January 2025, joint EU/UK packs can no longer be released into the UK market. When adding the ‘UK Only’ statement, all administrative details must be removed from the outer cartons of these packs. However, packs already released to the market may continue to be supplied until their expiry date. Shared inner packaging components, such as multilingual blister foils and joint leaflets, may still be used as long as UK and EU licences remain aligned. (A joint pack refers to packaging shared with one or more EU countries, displaying administrative details for both the UK and the other markets involved.)
UK only Stickering and limitation:
The ‘UK Only’ statement may be applied via a sticker for a limited period of six months, until 30th June 2025. After this date, ‘UK Only’ must be printed directly onto the packaging, as stickering will no longer be accepted. However, Parallel Imports (PLPI) products will remain unaffected by this guidance, and stickering and over-labelling will still be permitted for these products. Specific guidance on UK-only stickering has been provided by the agency, and it is recommended to review the directive’s specifications. For further details, please refer to the “Guidance for Wholesale Dealers and Manufacturers”.
To ensure the continued supply of medicines to Northern Ireland and for the sole purpose of adding ‘UK Only’ stickers to packs released prior to 1st January 2025, MA holders should use the notification process to name the Manufacturing Import Authorisation (MIA) holders (authorised for secondary packaging) that they intend to use and to support the notification for the responsibilities of QP are defined in EU GMP Annex 16 (1.4.1).
How to notify the agency of the change
The Marketing Authorisation Holder (MAH) can choose from the three options below to update the label in compliance with the Windsor Framework. Each option is subject to the applicable standard regulatory fee.
Any regulatory opportunity
An artwork change can be proposed alongside an ongoing or proposed variation (other than Type IA), with a statement in the cover letter confirming that the labelling has been updated in accordance with the Windsor Framework requirements.
Self-certification
A separate notification can be submitted for an artwork change. MAHs can implement the proposed changes once the application has been submitted and need not wait for formal approval.
Self-certification without initial Electronic Common Technical Document (eCTD)
The deadline for submitting artwork changes (notification) without an updated eCTD sequence was 31st December 2024. MAHs have until 31st December 2025 to submit an updated eCTD sequence.
Please refer the MHRA Windsor Framework guidance document for implementation.
Quick recap on implementing the Windsor Framework
- Implementation date for the Framework is 1st January 2025.
- Medicines with EU (centrally authorised products) will no longer be valid in NI.
- Medicines for the UK market must carry a “UK only” label with good legibility without masking any written or pictorial material.
- UK MHRA becomes the sole regulatory authority in NI.
The Windsor Framework will segregate medicines into one of two categories.
- Category 1 – The products will be authorized UK-wide under UK law and for pharmacovigilance, only UK requirements apply.
- Category 2 – All other products that will comply with applicable EU law and do not fall into mandatory scope of the centralised procedure. For pharmacovigilance, both UK and EU requirements are applicable.
- From 1st January 2025, new marketing authorisation applicants will be asked to assign a category for the submission and will remain fixed for the product lifecycle.
By consolidating the UK’s medicine approval process under MHRA and eliminating the complexities of EU oversight in Northern Ireland, the Windsor Framework creates a more predictable and unified market. However, with this transition comes the challenge of adapting swiftly to new labelling, packaging, and pharmacovigilance requirements. Those who act early will ensure seamless market access and enhance regulatory agility in a post Brexit landscape. The Windsor Framework is an opportunity to build a more resilient and responsive pharmaceutical regulatory system in the UK.
References
- The Protocol on Ireland and Northern Ireland explained - Consilium
- Northern Ireland Protocol: The Windsor Framework - House of Commons Library
- Labelling and packaging of medicinal products for human use following agreement of the Windsor Framework - GOV.UK
About the authors
 Mallikaarjunan R has 20+ years of experience in regulatory operations and manages Navitas Life Sciences’ regulatory services and technology. He brings rich experience in pharmaceutical, consumer and medical device portfolio having worked with top 10 pharmaceutical companies across the globe helping to develop/support regulatory strategies, operational improvement, programme management, predictive forecasting, develop automation/RPA, and resource plans to manage the supply and demand of the workforce.
Mallikaarjunan R has 20+ years of experience in regulatory operations and manages Navitas Life Sciences’ regulatory services and technology. He brings rich experience in pharmaceutical, consumer and medical device portfolio having worked with top 10 pharmaceutical companies across the globe helping to develop/support regulatory strategies, operational improvement, programme management, predictive forecasting, develop automation/RPA, and resource plans to manage the supply and demand of the workforce.
 Raghunandan Reddy has extensive experience in the regulatory domain and currently works for Navitas Life Sciences' regulatory compliance services and technology. He has a strong background in the pharmaceutical industry, having worked with leading pharmaceutical companies to develop and support regulatory strategies, ensure compliance management, drive operational improvements, and enhance product development.
Raghunandan Reddy has extensive experience in the regulatory domain and currently works for Navitas Life Sciences' regulatory compliance services and technology. He has a strong background in the pharmaceutical industry, having worked with leading pharmaceutical companies to develop and support regulatory strategies, ensure compliance management, drive operational improvements, and enhance product development.